First law of thermnodynamics is given by
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of gas with $\gamma=1.5$ is taken through an adiabatic process in which the volume is compressed from $1200\, {cm}^{3}$ to $300\, {cm}^{3}$. If the initial pressure is $200\, {kPa}$. The absolute value of the workdone by the gas in the process $= \,..... J.$View Solution
- 2View SolutionIn thermodynamics, heat and work are
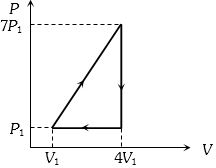
- 3View SolutionIn the cyclic process shown in the figure, the work done by the gas in one cycle is
- 4For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
- 5$540$ calories of heat converts $1$ cubic centimeter of water at $100\,^oC$ into $1671$ cubic centimeter of steam at $100\,^oC$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly .......... $cal$View Solution
- 6A monoatomic gas $\left( {\gamma = \frac{5}{3}} \right)$ is suddenly compressed to $\frac{1}{8}$ of its original volume, then the pressure of gas will change to how many times the initial pressure?View Solution
- 7During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
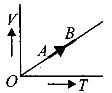
- 8The volume $( V)$ of a monatomic gas varies with its temperature $(T)$ , as shown in the graph. The ratio of work done by the gas , to the heat absorbed by it, when it undergoes a change from state $A$ to state $B$ , isView Solution
- 9$5.6$ liter of helium gas at $STP$ is adiabatically compressed to $0.7$ liter. Taking the initial temperature to be $T _1$, the work done in the process isView Solution
- 10A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
