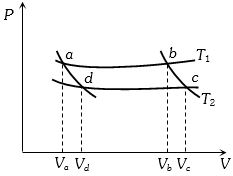
In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will be

Medium
(a) For adiabatic process ${T_1}V_b^{\gamma - 1}$= Constant
For $bc$ curve ${T_1}V_b^{\gamma - 1} = {T_2}V_c^{\gamma - 1}$ or $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{V_b}}}{{{V_c}}}} \right)^{\gamma - 1}}$…..$(i)$
For $ad$ curve ${T_1}V_a^{\gamma - 1} = {T_2}V_d^{\gamma - 1}$ or $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{V_a}}}{{{V_d}}}} \right)^{\gamma - 1}}$…..$(ii)$
From equation $(i)$ and $(ii)$ $\frac{{{V_b}}}{{{V_c}}} = \frac{{{V_a}}}{{{V_d}}}$
For $bc$ curve ${T_1}V_b^{\gamma - 1} = {T_2}V_c^{\gamma - 1}$ or $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{V_b}}}{{{V_c}}}} \right)^{\gamma - 1}}$…..$(i)$
For $ad$ curve ${T_1}V_a^{\gamma - 1} = {T_2}V_d^{\gamma - 1}$ or $\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{V_a}}}{{{V_d}}}} \right)^{\gamma - 1}}$…..$(ii)$
From equation $(i)$ and $(ii)$ $\frac{{{V_b}}}{{{V_c}}} = \frac{{{V_a}}}{{{V_d}}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
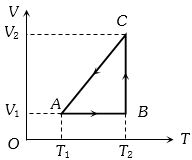
- 2A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectivelyView Solution
- 3View SolutionFirst law thermodynamics states that
- 4View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
- 5An ideal gas expands from volume $V_1$ to $V_2$. This may be achieved by either of the three processes: isobaric, isothermal and adiabatic. Let $\Delta U$ be the change in internal energy of the gas, $Q$ be the quantity of heat added to the system and $W$ be the work done by the system on the gas. Identify which of the following statements is false for $\Delta U$?View Solution
- 6Given that $1\,g$ of water in liquid phase has volume $1\,cm^3$ and in vapour phase $1671\, cm^3$ at atmospheric pressure and the latent heat of vaporization of water is $2256\,J/g;$ the change in the internal energy in joules for $1\,g$ of water at $373\,K$ when it changes from liquid phase to vapour phase at the same temperature is ....... $J$View Solution
- 7A mass of diatomic gas $(\gamma = 1 .4)$ at a pressure of $2$ atmospheres is compressed adiabatically so that its temperature rises from $27^o C$ to $927^o C.$ The pressure of the gas in the final state is ...... $atm$View Solution
- 8The gas law $\frac{{PV}}{T} = $ constant is true forView Solution
- 9Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansionView Solution
- 10A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
