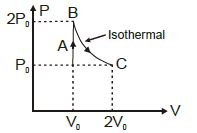
The figure shows $P-V$ diagram of a thermodynamic cycle. Which corresponding curve is correct?
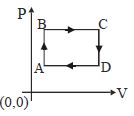

Medium
$\begin{array}{lll}\text { For } A-B: V=\text { constant } & \text { (ideal qas equation } \Rightarrow) \\ (P \propto T) & (\text { straight line) } & (P V=n R T \\ A B & & (P \alpha T) \text { for } A B\end{array}$
For $B-C \because P=$ constant $(v \propto T)$ for $B C$
$(V \propto T)$ (constant line parallel to $T($ temperature)) axis.
For $C D$ it will be similar to $A B$
and $DA$ It will be simicar to $BC$
$\therefore$ considering all above constraints option $(A)$ is the correct graph
For $B-C \because P=$ constant $(v \propto T)$ for $B C$
$(V \propto T)$ (constant line parallel to $T($ temperature)) axis.
For $C D$ it will be similar to $A B$
and $DA$ It will be simicar to $BC$
$\therefore$ considering all above constraints option $(A)$ is the correct graph
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
- 2A certain amount of gas is taken through a cyclic process $(A\,B\,C\,D\,A)$ that has two isobars, one isochore and one isothermal. The cycle can be represented on a $P-V$ indicator diagram asView Solution
- 3View SolutionIn isothermal expansion, the pressure is determined by
- 4Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 5$Assertion :$ In an isolated system the entropy increases.View Solution
$Reason :$ The processes in an isolated system are adiabatic. - 6During an adiabatic expansion of $2\, moles$ of a gas, the change in internal energy was found $-50J.$ The work done during the process is ...... $J$View Solution
- 7A cylinder of mass $1\,kg$ is given heat of $20000\, J$ at atmospheric pressure. If initially temperature of cylinder is $20\,^oC$, then work done by the cylinder will be .......$J$ (Given that Specific heat of cylinder $= 400 \,J\, kg^{-1}$, Coefficient of volume expansion $= 9 \times {10^{-5}}\,^o C^{-1}$, Atmospheric pressure $= 10^5 \,N/m^2$ and density of cylinder $9000\,kg/m^3$)View Solution
- 8One mole of an ideal gas is contained with in a cylinder by a frictionless piston and is initially at temperature $T$. The pressure of the gas is kept constant while it is heated and its volume doubles. If $R$ is molar gas constant, the work done by the gas in increasing its volume isView Solution
- 9View SolutionEntropy of a system decreases .........
- 10A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution