Molecules of an ideal gas are known to have three translational degrees of freedom and two rotational degrees of freedom. The gas is maintained at a temperature of $T$. The total internal energy, $U$ of a mole of this gas, and the value of $\gamma\left(=\frac{ C _{ P }}{ C _{ v }}\right)$ given, respectively, by
JEE MAIN 2020, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A container is filled with $20$ moles of an ideal diatomic gas at absolute temperature $T$. When heat is supplied to gas temperature remains constant but $8$ moles dissociate into atoms. Heat energy given to gas is .........View Solution
- 2For hydrogen gas ${C_p} - {C_v} = a$ and for oxygen gas ${C_p} - {C_v} = b$. So the relation between $a$ and $b$ is given byView Solution
- 3A gas mixture consists of molecules of type $1, 2$ and $3$, with molar masses ${m_1} > {m_2} > {m_3}.$ ${V_{rms}}$ and $\overline K $ are the $r.m.s.$ speed and average kinetic energy of the gases. Which of the following is trueView Solution
- 4Speed of sound in a gas is $v$ and $r.m.s.$ velocity of the gas molecules is $c.$ The ratio of $v$ to $c$ isView Solution
- 5The pressure of an ideal gas is written as $E = \frac{3PV}{2}$ . Here $E$ stands forView Solution
- 6A container with insulating walls is divided into two equal parts by a partition fitted with a valve. One part is filled with an ideal gas at a pressure $P$ and temperature $T$, whereas the other part is completely evacuated. If the valve is suddenly opened, the pressure and temperature of the gas will beView Solution
- 7Pressure versus temperature graph of an ideal gas at constant volume $V$ of an ideal gas is shown by the straight line $A$. Now mass of the gas is doubled and the volume is halved, then the corresponding pressure versus temperature graph will be shown by the lineView Solution
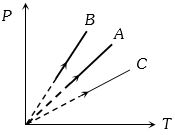
- 8According to the kinetic theory of gases the $r.m.s.$ velocity of gas molecules is directly proportional toView Solution
- 9A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
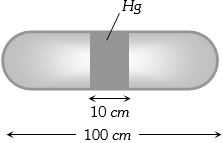
- 10The change in the magnitude of the volume of an ideal gas when a small additional pressure $\Delta P$ is applied at a constant temperature, is the same as the change when the temperature is reduced by a small quantity $\Delta T$ at constant pressure. The initial temperature and pressure of the gas were $300\, K$ and $2\; atm$ respectively. If $|\Delta T|=C|\Delta P|$ then value of $C$ in $(K / a t m)$ is......View Solution
