$N _{2}$ gas is heated from $300\, K$ temperature to $600\, K$ through an isobaric process. Then find the change in entropy of the gas. $( n =1 mole )$ (in $J/K$)
AIIMS 2019, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas undergoes an adiabatic process obeying the relation $PV^{4/3} =$ constant. If its initial temperature is $300\,\, K$ and then its pressure is increased upto four times its initial value, then the final temperature is (in Kelvin):View Solution
- 2A diatomic ideal gas is compressed adiabatically to $\frac{1}{32}$ of its initial volume. If the initial temperature of the gas is $T_1$ (in Kelvin) and the final temperature is $a T_1$, the value of $a$ isView Solution
- 3View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
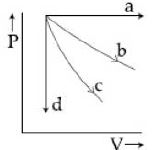
- 4In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
- 5$P-V$ plots for two gases during adiabatic process are shown in the figure. Plots $1$ and $2$ should correspond respectively toView Solution
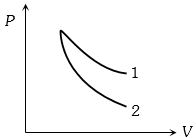
- 6The volume of a gas is reduced adiabatically to $\frac{1}{4}$ of its volume at $27°C$, if the value of $\gamma = 1.4,$ then the new temperature will beView Solution
- 7$1\,g$ of a liquid is converted to vapour at $3 \times 10^5\,Pa$ pressure. If $10 \%$ of the heat supplied is used for increasing the volume by $1600\,cm ^3$ during this phase change, then the increase in internal energy in the process will be $............\,J$View Solution
- 8View SolutionWhich of the following statements is correct for any thermodynamic system
- 9View SolutionAn ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
- 10$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope.
