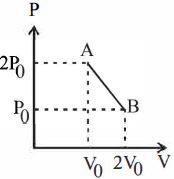
$P = \frac{{ - {P_0}}}{{{V_0}}}V + 3P$
$[slope = \frac{{ - {P_0}}}{{{V_0}}},c = 3{P_0}]$
$P{V_0} + {P_0}V = 3{P_0}{V_0}\,\,\,\,\,\,\,\,\,\,\,...\left( i \right)$
$But\,\,\,\,\,\,\,PV = nRT\,$
$\therefore P = \frac{{nRT}}{V}\,\,\,\,\,\,\,\,\,\,\,\,\,\,...\left( {ii} \right)$
$From\left( i \right)\& \left( {ii} \right)\frac{{nRT}}{V}{V_0} + {P_0}V = 3{P_0}{V_0}$
$\therefore nRT{V_0} + {P_0}{V^2} = 3{P_0}{V_0}$
$...\left( {iii} \right)$
For temperature to be maximum $\frac{{dT}}{{dV}} = 0$
Differentiating $e.q.(iii)\,by\,'v'\,we\,get$
$nR{V_0}\frac{{dT}}{{dV}} + {P_0}\left( {2v} \right) = 3{P_0}{V_0}$
$\therefore nR{V_0}\frac{{dT}}{{dV}} = 3{P_0}{V_0} - 2{P_0}V$
$\frac{{dT}}{{dV}} = \frac{{3{P_0}{V_0} - 2{P_0}V}}{{nR{V_0}}} = 0$
$V = \frac{{3{V_0}}}{2}\,\,\,\,\,\,\,\,\,\therefore P = \frac{{3{P_0}}}{2}$ $[From (i)]$
$\therefore \,{T_{\max }} = \frac{{9{P_0}{V_0}}}{{4nR}}\,\,\left[ {From\,\left( {iii} \right)} \right]$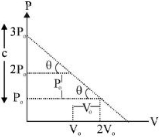
Download our appand get started for free
Similar Questions
- 1For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
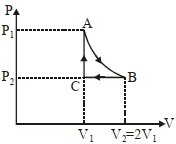
- 2$n$ mole a perfect gas undergoes a cyclic process $ABCA$ (see figure) consisting of the following processes.View Solution
$A \rightarrow B :$ Isothermal expansion at temperature $T$ so that the volume is doubled from $V _{1}$ to $V _{2}=2 V _{1}$ and pressure changes from $P _{1}$ to $P _{2}$
$B \rightarrow C :$ Isobaric compression at pressure $P _{2}$ to initial volume $V _{1}$
$C \rightarrow A$ : Isochoric change leading to change of pressure from $P _{2}$ to $P _{1}$
Total workdone in the complete cycle $ABCA$ is
- 3A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
- 4A perfect gas of a given mass is heated first in a small vessel and then in a large vessel, such that their volumes remain unchanged. The $P-T$ curves areView Solution
- 5A certain amount of gas is taken through a cyclic process $(A\,B\,C\,D\,A)$ that has two isobars, one isochore and one isothermal. The cycle can be represented on a $P-V$ indicator diagram asView Solution
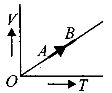
- 6The volume $( V)$ of a monatomic gas varies with its temperature $(T)$ , as shown in the graph. The ratio of work done by the gas , to the heat absorbed by it, when it undergoes a change from state $A$ to state $B$ , isView Solution
- 7Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
- 8A solid body of constant heat capacity $1\ J/^o C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with $8$ reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^o C$ to final temperature $200^o C$. Entropy change of the body in the two cases respectively is :
- 9View SolutionFor an isometric process
- 10In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20\,J$ of heat and $8\,J$ of work is done on the gas. If the initial internal energy of the gas was $30\,J$. The final internal energy will be...... $J$View Solution
