For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):
JEE MAIN 2021, Medium
$PV ^{\gamma}=$ constant
Differentiating
$\frac{ dP }{ dV }=-\frac{\gamma P }{ V }$
$\frac{ dP }{ P }=-\frac{\gamma dV }{ V }$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
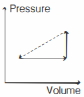
- 1One mole of an ideal gas goes from an initial state $A$ to final state $B$ via two processes : It first undergoes isothermal expansion from volume $V$ to $3\, V$ and then its volume is reduced from $3\, V$ to $V$ at constant pressure. The correct $P-V$ diagram representing the two processes isView Solution
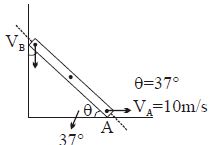
- 2Find $V_B = ?$View Solution
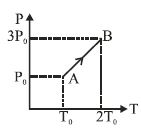
- 3Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
- 4A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
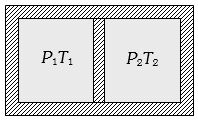
- 5Following figure shows on adiabatic cylindrical container of volume ${V_0}$ divided by an adiabatic smooth piston (area of cross-section = $A$ ) in two equal parts. An ideal gas $({C_P}/{C_V} = \gamma )$ is at pressure $P_1$ and temperature $T_1$ in left part and gas at pressure $P_2$ and temperature $T_2$ in right part. The piston is slowly displaced and released at a position where it can stay in equilibrium. The final pressure of the two parts will be (Suppose $ x$ = displacement of the piston)View Solution
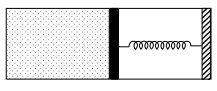
- 6An ideal monoatomic gas is confined in a horizontal cylinder by a spring loaded piston (as shown in the figure). Initially the gas is at temperature $T _1$, pressure $P_1$ and volume $V_1$ and the spring is in its relaxed state. The gas is then heated very slowly to temperature $T_2$, pressure $P _2$ and volume $V _2$. During this process the piston moves out by a distance $x$. Ignoring the friction between the piston and the cylinder, the correct statement$(s)$ is(are)View Solution
$(A)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the energy stored in the spring is $\frac{1}{4} P_1 V_1$
$(B)$ If $V_2=2 V_1$ and $T_2=3 T_1$, then the change in internal energy is $3 P_1 V_1$
$(C)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the work done by the gas is $\frac{7}{3} P_1 V_1$
$(D)$ If $V_2=3 V_1$ and $T_2=4 T_1$, then the heat supplied to the gas is $\frac{17}{6} P_1 V_1$
- 7View SolutionThe internal energy of the gas increases In
- 8A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
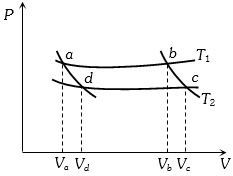
- 9In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
- 10An ideal gas is made to undergo the cyclic process shown in the figure below. Let $\Delta W$ depict the work done, $\Delta U$ be the change in internal energy of the gas and $Q$ be the heat added to the gas. Sign of each of these three quantities for the whole cycle will be (0 refers to no change)View Solution