$\therefore \,dQ = n{C_p}dT;\,where\,{C_p}\,is\,specific\,heat\,at\,constant\,pressure.$
or $dQ = n\left( {\frac{5}{2}R} \right)dT$
$Also,\,dW = PdV = nRdT\,\left( {PV = nRT} \right)$
Required ratio$ = \frac{{dW}}{{dQ}} = \frac{{nRdT}}{{n\left( {\frac{5}{2}R} \right)dT}} = \frac{2}{5}$
Download our appand get started for free
Similar Questions
- 1An engine has an efficiency of $0.25$ when temperature of sink is reduced by $58\,^oC$, if its efficiency is doubled, then the temperature of the source is ..... $^oC$View Solution
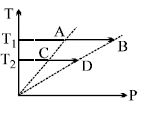
- 2On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
- 3A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 4A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 5A diatomic gas initially at $18^o C$ is compressed adiabatically to one-eighth of its original volume. The temperature after compression will beView Solution
- 6A heat engine operates with the cold reservoir at temperature $324 K$. The minimum temperature of the hot reservoir, if the heat engine takes $300 \; J$ heat from the hot reservoir and delivers $180 \; J$ heat to the cold reservoir per cycle, is $\dots \; K .$View Solution
- 7An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
- 8A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
- 9View SolutionA cycle tyre bursts suddenly. This represents an
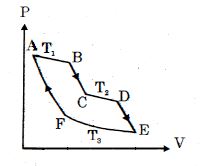
- 10One mole of ideal gas taken through a cycle process with alternate isothermal and adiabatic curves. In $P-V$ diagram $AB, CD, EF$ are isothermal curves at the absolute temperature $T_1, T_2$ and $T_3$ respectively and $BC, DE$ and $FA$ are adiabatic curves respectively. If $\frac{{{V_B}}}{{{V_A}}} = 2,\,\frac{{{V_D}}}{{{V_C}}} = 2$ then for cycle is shown in figure four statements are being made given below. (Figure is not drawn on scale)View Solution
Statement $1$ : Ratio of volumes $\frac{{{V_E}}}{{{V_F}}} = 4$
Statement $2$ : Magnitude of work done in isothermal compression $EF$ is $2RT_3\ ln\ (2)$
Statement $3$ : Ratio of heat supplied to gas in the process $AB$ to heat rejected by gas in process $EF$ is $\frac{{{T_1}}}{{2{T_3}}}$
Statement $4$ : Net work done by gas in the cycle $ABCDEFA$ is $(T_1 + T_2 - 2T_3) R\ ln\ (2)$
Find the number of correct statement $(s)$ given for the cyclic process followed by gas