A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1During the melting of a slab of ice at $273\, K$ at atmospheric pressureView Solution
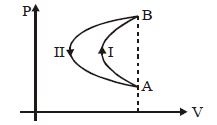
- 2In a cyclic process, a gas is taken from state $A$ to $B$ via path $-I$ as shown in the indicator diagram and taken back to state $A$ from state $B$ via path $-II$ . In the complete cycleView Solution
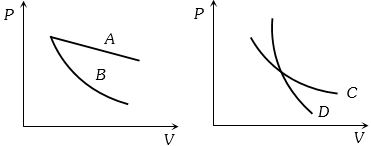
- 3In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution
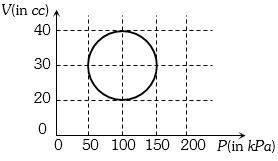
- 4View SolutionA system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is
- 5$5.6$ $liter$ of helium gas at $STP$ is adiabatically compressed to $0.7$ $liter$. Taking the initial temperature to be $T_1$, the work done in the process isView Solution
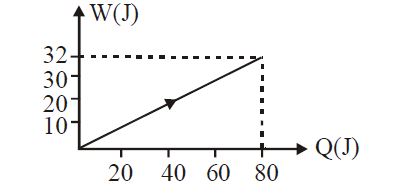
- 6View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
- 7View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
- 8An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 9$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution
- 10An ideal Carnot heat engine with an efficiency of $30\%$.It absorbs heat from a hot reservoir at $727^o C$. The temperature of the cold reservoir is .... $^oC$View Solution
