Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View Solutionપ્રથમ પાંચ સંક્રાંતિ તત્વોના ક્રમિક ચાર સભ્યો તેમની અણુ સંખ્યા સાથે નીચે સૂચિબદ્ધ છે. તેમાંથી કયામાં સૌથી વધુ ત્રીજી આયનીકરણ થવાની સંભાવના છે
- 2View Solutionધાતુ ધનાયનનો ઓક્સાઇડ જે ઊભયગુણી નથી?
- 3નીચે આપેલામાંથી $\mathrm{d}^{10}$ ઈલેક્ટ્રોન સંરચના ધરાવતા બધા જ તત્વો માટેનો સાચો વિકલ્પ પસંદ કરો.View Solution
- 4સંકિર્ણ $[M(H_2O)_6]Cl_2$ માં ધાતુ આયનની કઈ જોડી $3.9\, BM$ ની ફક્ત સ્પિન ચુંબકીય ચાકમાત્રા આપે છે?View Solution
- 5સંક્રાતિ ધાતુ આયનની ચુંબકીની ચુંબકીય ચાકમાત્રા $3.87\,B.M$ તેમાં કેટલા અયુગ્મીત ઇલેકટ્રોનની સંખ્યા હજર હોય છે.View Solution
- 6View Solutionનીચેનામાંથી કયુ સંક્રાતિ ધાતુ આયન નિશ્વિત ચુંબકીય ચાકમાત્રા ધરાવે છે ?
- 7View Solutionલેન્થેનાઈડ સંકોચન શા કારણે પરિણમે છે?
- 8ધાતુના આયન $M^{x+}$ $(Z = 24)$ ની સંયોજનમાં બોન મેગ્નેટન્સનો સ્પિન ફક્ત ચુંબકીય ક્ષણ છે સંયોજન માં અબંધકારક ઇલેક્ટ્રોન ની સંખ્યા કેટલી હશે ?View Solution
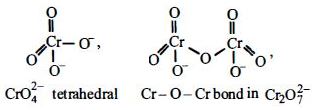
- 9$4{K_2}C{r_2}{O_7}\xrightarrow{{{\text{heat}}}}4{K_2}Cr{O_4} + 3{O_2} + X$ ઉપરની પ્રક્રિયાઓમાંએ .................. છે.View Solution
- 10નીચે આપેલા લેન્થેનોઈડ્સસ પૈકી એક $+4$ ઓક્સિડેશન અવસ્થા દર્શાવી ઉમદા વાયુની ૨ચના (વિન્યાસ) પ્રાપ્ત કરી શકે ?View Solution
(૫રમામાણ્વીય ક્રમાંક : $La =57, Ce =58, Eu =63$ અને $Yb =70)$