$\Delta H_f^o\left( {CO} \right) = - 110.5\,kJ\,mo{l^{ - 1}};$
$\Delta H_f^o\left( {C{O_2}} \right) = - 393.5\,kJ\,mo{l^{ - 1}}$
\(NO + CO \longrightarrow \frac{1}{2} N _2+ CO _2\)
\(\Delta H ^{\circ}=\left|\frac{1}{2} \Delta_1 H _{ N _2}^{\circ}+2 \Delta_1 H _{ CO _2}^{\circ}\right|-\left|\Delta_1 H _{ NO }^{\circ}+\Delta_1 H _{ CO }^{\circ}\right|\)
\(-372.2=\left|\frac{1}{2}(0)+(-393.5)\right|-\left|\Delta_1 H _{ NO }^0+(-110.5)\right|\)
\(\therefore \Delta_1 H _{ NO }^{\circ}=-393.5+110.5+372.2=-393.5+482.7\)
\(=89.2\, kJ\, mol ^{-1}\)
Download our appand get started for free
Similar Questions
- 1ઉષ્મા એન્જિન તાપમાન ${T_1}$ પર ઉષ્મા ${Q_1}$ શોષે છે અને તાપમાન ${T_2}$ પર ${Q_2}$ ઉષ્મા શોષી લે છે. એન્જિન દ્વારા કરવામાં આવેલ કાર્ય $({Q_1} + {Q_2})$ છે. આ માહિતિ પરથી,..View Solution
- 2એક મોલ પ્રવાહી સ્વરૂપ પાણીનું વરાળમાં રૂપાંતર કરતા $373 \,K$ તાપમાને એન્ટ્રોપી ફેરફાર ...... જૂલ/કેલ્વિન ($\Delta S$ (બાષ્પાયન) $= 2.257$ કિલોજૂલ/ગ્રામ)View Solution
- 3View Solutionઅચળ દબાણમાં બરફ સાથે સંતુલનમાં પાણીની મોલર ઉષ્મા ક્ષમતા છે
- 4નીચે બે પ્રક્રિયાઓ આપેલ છે.View Solution
$2 \mathrm{Fe}_{(\mathrm{s})}+\frac{3}{2} \mathrm{O}_{2(\mathrm{~g})} \rightarrow \mathrm{Fe}_2 \mathrm{O}_{3(\mathrm{~s})}, \Delta \mathrm{H}^{\mathrm{o}}=-822 \mathrm{~kJ} / \mathrm{mol}$
$\mathrm{C}_{(\mathrm{s})}+\frac{1}{2} \mathrm{O}_{2(\mathrm{~g})} \rightarrow \mathrm{CO}_{(\mathrm{g})}, \Delta \mathrm{H}^{\mathrm{o}}=-110 \mathrm{~kJ} / \mathrm{mol}$
$3\mathrm{C}_{(\mathrm{s})}+\mathrm{Fe}_2 \mathrm{O}_{3(\mathrm{~s})} \rightarrow 2 \mathrm{Fe}_{(\mathrm{s})}+3 \mathrm{CO}_{(\mathrm{g})}$ આપેલા પ્ર્ક્રિયા માટે એન્થાલ્પી ફેરફાર__ _ _$J/mol$ છે.
- 5એક બોમ્બ કેલોરીમીટરમાં, સાયનામાઈડ $NH _{2} CN _{( s )}$ અને ઓકિસજનની પ્રક્રિયા કરવામાં આવે છે અને તેનો $\Delta U -742.24\, kJ\, mol ^{-1}$ માલૂમ પડયો. તો આપેલી પ્રક્રિયા માટે,View Solution
$NH _{2} CN _{( s )}+\frac{3}{2} O _{2}( g ) \rightarrow N _{2( g )}+ O _{2}( g )+ H _{2} O _{(l)}$
$\Delta H _{298}$ ની માત્રા ........ $kJ$ છે. (નજીક પૂર્ણાંક રાઉન્ડ ઓફ)
[ધારી લો આદર્શ વાયુઓ અને $\left. R =8.314\, J\, mol ^{-1} K ^{-1}\right]$
- 6$25^o$ બોમ્બ કેલેરી મીટરમાં ઈથેનોલની દહન ઉષ્મા $-670.48\, K.$ કેટલી મોલ$^{-1}$ છે. તો આ પ્રક્રિયા માટે $25\,^oC$ એ $\Delta$$H$ નું મૂલ્ય કેટલા .....$K.\, Cals.$ ?View Solution
- 7ઓટોમોબાઈલમાં થતી નીચેની પ્રક્રિયા માટે $\Delta H, \Delta S, \Delta G$ નાં મૂલ્યો અનુક્રમે ...... હોય છે.View Solution
$2C_8H_{18}$ $_{(g)}$ + $25O_2$ $_{(g)}$ $\rightarrow$ $16CO_2$$_{(g)}$ + $18H_2O$ $_{(g)}$
- 8$15.5\, g$ પ્રોપેનના દહનથી કેટલા $.......kJ$ ઉષ્મા ઉત્પન્ન થાય ? ${C_3}{H_8} + 5{O_2} \to 3C{O_2} + 4{H_2}O\,;\,\Delta {H^o} = - 2219\,kJ/mol$View Solution
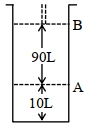
- 9આપેલ આક્રૂતિને ધ્યાનમાં લો.View Solution
$18^{\circ} \mathrm{C}$ પર, સ્થાન $A$ પર, પિસ્ટન સાથે જોડેલા (fitted) સિલિન્ડર માં આદર્શ વાયુનો $1$ $\mathrm{mol}$ રાખેલ છે. જો તાપમાન માં કોઈપણ જાતનો ફેરફાર ન કરીએ તો પિસ્ટન એ સ્થાન $B$ તરફ ખસે છે ત્યારે આ પ્રતિવર્તી પ્રક્રમ માં થયેલ કાર્ય $'x' L atm$ છે. $x=-$ ........... $L.atm$ (નજીક નો પૂર્ણાક)
[આપેલ : નિરપેક્ષ તાપમાન $={ }^{\circ} \mathrm{C}+273.15, \mathrm{R}=0.08206 \mathrm{~L} \mathrm{~atm} \mathrm{~mol}^{-1} \mathrm{~K}^{-1}$ ]
- 10View Solutionકઈ પ્રક્રિયામાં એન્ટ્રોપી વધે છે ?
