Nitrogen gas is filled in an insulated container. If $\alpha$ fraction of moles dissociates without exchange of any energy, then the fractional change in its temperature is ..............
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If the intermolecular forces vanish away, the volume occupied by the molecules contained in $4.5 \,kg$ water at standard temperature and pressure will beView Solution
- 2Hydrogen gas is filled in a balloon at $20^\circ C$. If temperature is made $40^\circ C$, pressure remaining same, what fraction of Hydrogen will come outView Solution
- 3A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300\ K.$ The ratio of the average rotational kinetic energy per $O_2$ molecule to that per $N_2$ molecule isView Solution
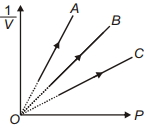
- 4Figure shows the isotherms of a fixed mass of an ideal gas at three temperatures $T_A, T_B$ and $T_C$, thenView Solution
- 5$E_0$ and $E_H$ respectively represents the average kinetic energy of a molecule of oxygen and hydrogen. If the two gases are at the same temperature, which of the following statement is true?View Solution
- 6If $10^{22}$ gas molecules each of mass $10^{-26}\, kg$ collide with a surface (perpendicular to it)elastically per second over an area $1\, m^2$ with a speed $10^4\,m/s$, the pressure exerted by the gas molecules will be of the order ofView Solution
- 7Open container contains $x$ mole of gas at $T$ $kelvin$ If temperature is tripled, then the amount of gas exit the container isView Solution
- 8During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its absolute temperature. The ratio of $\frac{C_p}{C_v}$ for the gas is:View Solution
- 9If one mole of the polyatomic gas is having two vibrational modes and $\beta$ is the ratio of molar specific heats for polyatomic gas $\left(\beta=\frac{ C _{ P }}{ C _{ v }}\right)$ then the value of $\beta$ is:View Solution
- 10View SolutionThe root mean square speed of the molecules of a gas is
