The root mean square speed of the molecules of a gas is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Statement$-1 :$ Internal energy of gas $U = nC_VT$ is due to random motion of gas molecules.View Solution
Statement$-2 :$ A container is moving with speed $v$. It is suddenly stopped by a force, temperature of gas increases. - 2In Maxwell's speed distribution curve, for $N_2$ gas, the average of $\mid$ relative velocity (in $m/s$) $\mid$ between two molecules at $300 \,K$ will beView Solution
- 3When the pressure on $1200\, ml$ of a gas in increased from $70\, cm $ to $120\, cm$ of mercury at constant temperature, the new volume of the gas will be ........ $ml$View Solution
- 4Which statements are correct about degrees of freedom?View Solution
$A.$ $A$ molecule with $n$ degrees of freedom has $n^{2}$ different ways of storing energy.
$B.$ Each degree of freedom is associated with $\frac{1}{2} RT$ average energy per mole.
$C.$ $A$ monoatomic gas molecule has $1$ rotational degree of freedom where as diatomic molecule has $2$ rotational degrees of freedom
$D$ $CH _{4}$ has a total to $6$ degrees of freedom Choose the correct answer from the option given below:
- 5The temperature of a gas is $-78^{\circ} \mathrm{C}$ and the average translational kinetic energy of its molecules is $\mathrm{K}$. The temperature at which the average translational kinetic energy of the molecules of the same gas becomes $2 \mathrm{~K}$ is :View Solution
- 6If an ideal gas has volume $V$ at $27°C$ and it is heated at a constant pressure so that its volume becomes $1.5V.$ Then the value of final temperature will be ....... $^oC$View Solution
- 7Calculate the value of mean free path $(\lambda)$ for oxygen molecules at temperature $27^{\circ}\, C$ and pressure $1.01 \times 10^{5} \,Pa$. Assume the molecular diameter $0.3 \,nm$ and the gas is ideal. $\left( k =1.38 \times 10^{-23}\, J\,K ^{-1}\right)$ (in $nm$)View Solution
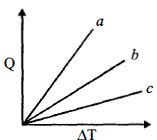
- 8Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond toView Solution
- 9Two identical adiabatic vessels are filled with oxygen at pressure $P_1$ and $P_2 (P_1 > P_2).$ The vessels are interconnected with each other by a nonconducting pipe. If $U_{01}$ and $U_{02}$ denote initial internal energy of oxygen in first and second vessel respectively and $U_{f_1}$ and $U_{f_2}$ denote final internal energy values, than :View Solution
- 10View SolutionA container with rigid walls is covered with perfectly insulating material. The container is divided into two parts by a partition. One part contains a gas while the other is fully evacuated (vacuum). The partition is suddenly removed. The gas rushes to fill the entire volume and comes to equilibrium after a little time. If the gas is not ideal, then
