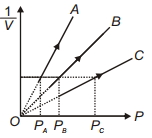
Figure shows the isotherms of a fixed mass of an ideal gas at three temperatures $T_A, T_B$ and $T_C$, then

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Molar specific heat of oxygen at constant pressure ${C_p} = 7.2\,cal/mol^\circ C$ and $R = 8.3 joule/mol/K$. At constant volume, $5\, mol$ of oxygen is heated from $10°C$ to $20°C$, the quantity of heat required is approximately ........ $cal$View Solution
- 2View SolutionIf two moles of diatomic gas and one mole of mono atomic gas are mixed then the ratio of specific heats is
- 3One litre of oxygen at a pressure of $1\, atm$ and two litres of nitrogen at a pressure of $0.5\, atm$ are introduced into a vessel of volume $1L$. If there is no change in temperature, the final pressure of the mixture of gas (in $atm$) isView Solution
- 4The curve between absolute temperature and ${v^2}_{rms}$ isView Solution
- 5The kinetic energy of one gram molecule of a gas at normal temperature and pressure is $(R = 8.31\,J/mole - K)$View Solution
- 6An inverted bell lying at the bottom of a lake $47.6\,m$ deep has $50\, cm^3$ of air trapped in it. The bell is brought to the surface of the lake. The volume of the trapped air will be ...... $cm^3$ (atmospheric pressure $= 70\, cm$ of $Hg$ and density of $Hg = 13.6\, g/cm^3$)View Solution
- 7Relationship between $P,V,$ and $E$ for a gas isView Solution
- 8Let $A$ and $B$ the two gases and given : $\frac{{{T_A}}}{{{M_A}}} = 4.\frac{{{T_B}}}{{{M_B}}};$ where $T$ is the temperature and M is molecular mass. If ${C_A}$ and ${C_B}$ are the $r.m.s. $ speed, then the ratio $\frac{{{C_A}}}{{{C_B}}}$ will be equal toView Solution
- 9The kinetic energy, due to translational motion, of most of the molecules of an ideal gas at absolute temperature $T$ isView Solution
- 10A gas at $27^o C$ has a volume $V$ and pressure $P.$ On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ...... $^oC$View Solution