If $Q , E$ and $W$ denote respectively the heat added, change in internal energy and the work done by a closed cycle process, then
AIPMT 2008, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
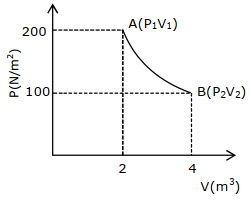
- 1A certain amount of gas of volume $V$ at $27^{o}\,C$ temperature and pressure $2 \times 10^{7} \;Nm ^{-2}$ expands isothermally until its volume gets doubled. Later it expands adiabatically until its volume gets redoubled. The final pressure of the gas will be (Use $\gamma=1.5$ )View Solution
- 2View SolutionWhich one is the correct option for the two different thermodynamic processes ?
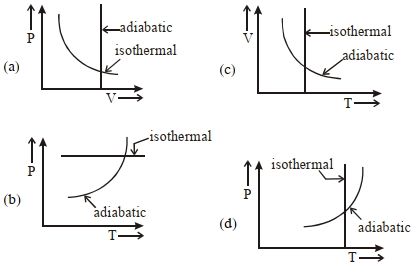
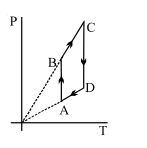
- 3View SolutionPressure versus temperature graph of an ideal gas is shown in figure
- 4$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
- 5Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
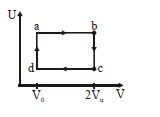
- 6The figure given below shows the variation in the internal energy $U$ with volume $V$ of $2.0\ mole$ of an ideal gas in a cyclic process $abcda$ . The temperatures of the gas during the processes $ab$ and $cd$ are $500\ K$ and $300\ K$ respectively, the heat absorbed by the gas during the complete process is .... $J$View Solution
(Take $R$ = $8.3\ J/mol-K$ and $ln\ 2$ = $0.69$)
- 7One mole of an ideal gas at $27^{\circ} {C}$ is taken from ${A}$ to ${B}$ as shown in the given ${PV}$ indicator diagram. The work done by the system will be $......\times 10^{-1} \,{J}$View Solution
[Given : $R=8.3\, {J} /\,mole\,{K}, \ln 2=0.6931$ ] (Round off to the nearest integer)
- 8The $PV$ diagram shows four different possible reversible processes performed on a monatomic ideal gas. Process $A$ is isobaric (constant pressure). Process $B$ is isothermal (constant temperature). Process $C$ is adiabatic. Process $D$ is isochoric (constant volume). For which process(es) does the temperature of the gas decrease ?View Solution
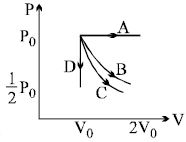
- 9One mole of helium is adiabatically expanded from its initial state $({P_i},{V_i},{T_i})$ to its final state $({P_f},{V_f},{T_f})$. The decrease in the internal energy associated with this expansion is equal toView Solution
- 10The temperature of $3.00\, {mol}$ of an ideal diatomic gas is increased by $40.0^{\circ} {C}$ without changing the pressure of the gas. The molecules in the gas rotate but do not oscillate. If the ratio of change in internal energy of the gas to the amount of workdone by the gas is $\frac{{x}}{10} .$ Then the value of ${x}$ (round off to the nearest integer) is ..... . $\left(\right.$ Given $\left.{R}=8.31\, {J} {mol}^{-1} {K}^{-1}\right)$View Solution
