Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansion
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal monoatomic gas is taken along the path $ABCA$ as shown in the $PV$ diagram. The maximum temperature attained by the gas along the path $BC$ is given byView Solution
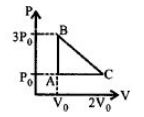
- 2An engine operating between the boiling and freezing points of water will haveView Solution
$1.$ efficiency more than $27 \%$
$2.$ efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
$3.$ efficiency equal to $27 \%$
$4.$ efficiency less than $27 \%$
- 3A monoatomic gas at pressure $P$ and volume $V$ is suddenly compressed to one eighth of its original volume. The final pressure at constant entropy will be $.....P$View Solution
- 4Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
- 5View SolutionThe work done in an adiabatic change in a gas depends only on
- 6If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
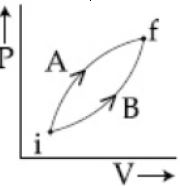
- 7Following figure shows two processes $A$ and $B$ for a gas. If $\Delta Q_A$ and $\Delta Q_B$ are the amount of heat absorbed by the system in two cases, and $\Delta U_A$ and $\Delta U_B$ are changes in internal energies, respectively, thenView Solution
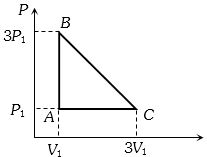
- 8An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
- 9A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 10View SolutionFor an ideal gas, in an isothermal process
