Which of the following statement($s$) is(are) correct?
$(A)$ The magnitude of the total work done in the process $A \rightarrow B \rightarrow C$ is $144 kJ$.
$(B)$ The magnitude of the work done in the process $B \rightarrow C$ is $84 kJ$.
$(C)$ The magnitude of the work done in the process $A \rightarrow B$ is $60 kJ$.
$(D)$ The magnitude of the work done in the process $C \rightarrow A$ is zero.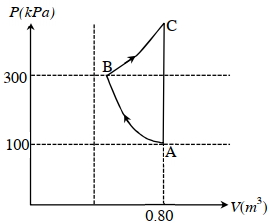
$P_A V_x^t=P_x V_x^t$
$10^2 \times(0.8)^{\frac{2}{2}}=3 \times 10^2\left(V_n\right)^{\frac{2}{1}}$
$\Rightarrow V_x=0.8 \times\left(\frac{1}{3}\right)^{0.2}=0.4$
Wodk doue in process $A \rightarrow B$
$W_{c x}=\frac{P_s V_0-P_x V_x}{\gamma-1}$
$\Rightarrow W_{\text {sx }}=\frac{10^2 \times 0.8-3 \times 10^2 \times 0.4}{\frac{5}{3}-1}$
$\Rightarrow W_{\text {As }}=-60 lJ =\Rightarrow\left|W_{\lambda \Omega}\right|=60 lJ$
Work done in process $B \rightarrow C$ (Isothermal process)
$W_{x=}=n R T / n \frac{V_8}{V_x}=P_x V_x \ell m \frac{V_8}{V_x}$
$\Rightarrow W_{x c}=3 \times 10^2 \times 0.4 \ln \frac{0.8}{0.4}$
$\Rightarrow W_{s c}=34 kJ$
Wodk doue in process $C \rightarrow A$
$W_{C_A}=P \Delta V=0 \quad(\because \Delta V=0)$
So toral work done in the process $A \rightarrow B \rightarrow C$
$W_{A B C}=W_{A \triangle}+W_{y C}+W_{C A}=-60+84+0$
$W_{A B C}=24 kJ$
So comect options are $(B,C.D)$
Download our appand get started for free
Similar Questions
- 1View SolutionWhich of the following statements is correct for any thermodynamic system
- 2$P-V$ plots for two gases during adiabatic process are shown in the figure. Plots $1$ and $2$ should correspond respectively toView Solution
- 3View SolutionIn a cyclic process, the internal energy of the gas
- 4Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$View Solution
- 5In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution
- 6A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
- 7Two Carnot engines $A$ and $B$ are operated in series. The first one, $A,$ receives heat at $T_1(= 600\,K)$ and rejects to a reservoir at temperature $T_2.$ The second engine $B$ receives heat rejected by the first engine and, in turns, rejects to a heat reservoir at $T_3 (=400\,K).$ Calculate the temperature $T_2$ if the work outputs of the two engines are equal ..... $K$View Solution
- 8A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
- 9Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansionView Solution
- 10View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?