One mole of a perfect gas in a cylinder fitted with a piston has a pressure $P,$ volume $V$ and temperature $T.$ If the temperature is increased by $1 \,K$ keeping pressure constant, the increase in volume is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
- 2When $1\, kg$ of ice at $0^o C$ melts to water at $0^o C,$ the resulting change in its entropy, taking latent heat of ice to be $80\, cal/gm,$ is ...... $cal/K$View Solution
- 3View SolutionWhich of the following is not a thermodynamics co-ordinate
- 4When $1\, kg$ of ice at $0^o C$ melts to water at $0^o C,$ the resulting change in its entropy, taking latent heat of ice to be $80\, cal/gm,$ is ...... $cal/K$View Solution
- 5View SolutionThe work done in an adiabatic change in a gas depends only on
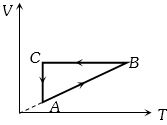
- 6A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
- 7View SolutionA gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half. Then
- 8$Assertion :$ The Carnot cycle is useful in understanding the performance of heat engines.View Solution
$Reason :$ The Carnot cycle provides a way of determining the maximum possible efficiency achievable with reservoirs of given temperatures. - 9View SolutionFirst law thermodynamics states that
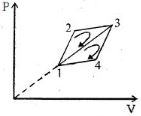
- 10The efficiency of a thermodynamic cycle $1-2-3- 1 ($see picture$)$ is $20\%$ and for another thermodynamic cycle $1 - 3-4 - 1$ efficiency is equal to $10\%$. Determine the efficiency $\eta ($ in $\%)$ of the thermodynamic cycle $1-2-3-4- 1.$The gas is assumed to be idealView Solution