One mole of an ideal diatomic gas is taken through the cycle as shown in the figure.
$1 \rightarrow 2 :$ isochoric process
$2 \rightarrow 3 :$ straight line on $P-V$ diagram
$3 \rightarrow 1 :$ isobaric process
The average molecular speed of the gas in the states $1, 2$ and $3$ are in the ratio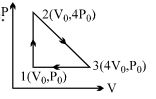
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An open and wide glass tube is immersed vertically in mercury in such a way that length $0.05\,\, m$ extends above mercury level. The open end of the tube is closed and the tube is raised further by $0.43 \,\,m$. The length of air column above mercury level in the tube will be ...... $m$ Take $P_{atm} = 76 \,\,cm$ of mercuryView Solution
- 2A box contains n molecules of a gas. How will the pressure of the gas be effected, if the number of molecules is made $2n$View Solution
- 3The kinetic energy of one gm-mole of a gas at normal temperature and pressure is $(R = 8.31 J/Mole-K)$View Solution
- 4Two moles of ideal helium gas are in a rubber balloon at $30^o C$. The balloon is fully expandable and can be assumed to required no energy in its expansion. The temperature of the gas in the balloon is slowly changed to $35^o C$. The amount of heat required in raising the temperature is nearly...... $J$View Solution
(take $R$ $=$ $8.31$ $J/mol.K$) - 5Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ The equivalent degree of freedom $f$ of the mixture isView Solution
- 6Hydrogen gas is filled in a balloon at $20^\circ C$. If temperature is made $40^\circ C$, pressure remaining same, what fraction of Hydrogen will come outView Solution
- 7$N( < 100)$ molecules of a gas have velocities $1, 2, 3\,.\,.\,.\,.\,.N/km/s$ respectively. ThenView Solution
- 8Three vessels of equal volume contain gases at the same temperature and pressure. The first vessel contains neon (monoatomic), the second contains chlorine (diatomic) and third contains uranium hexafloride (polyatomic). Arrange these on the basis of their root mean square speed $\left(v_{ ms }\right)$ and choose the correct answer from the options given below:View Solution
- 9A gas at $ 27\ ^oC$ has a volume $V$ and pressure $P$. On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ....... $^oC$View Solution
- 10If $10^{22}$ gas molecules each of mass $10^{-26}\, kg$ collide with a surface (perpendicular to it)elastically per second over an area $1\, m^2$ with a speed $10^4\,m/s$, the pressure exerted by the gas molecules will be of the order ofView Solution
