Given below are two statements:
Statement $I:$ If heat is added to a system, its temperature must increase.
Statement $II:$ If positive work is done by a system in a thermodynamic process, its volume must increase.
In the light of the above statements, choose the correct answer from the options given below
JEE MAIN 2023, Medium
Statement $I:$ $\Delta Q > 0$
According to $1^{\text {st }}$ law of thermodynamics
$\Delta Q =\Delta U + W$
If $\Delta Q > 0, \Delta U < 0$ and $W > 0$ is also possible.
Hence $\Delta T < 0$, so $T$ decreases.
Statement $I$ is false
Statement $II:$ W $ > 0$
$\therefore \int Pdv > 0$
Therefore volume of system must increase during positive work done by the system.
Statement $II$ is true
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed toView Solution
- 2Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
- 3Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
- 4View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter
- 5The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
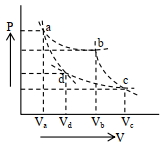
- 6Two different adiabatic paths for the same gas intersect two isothermal curves as shown in$P-V$ diagram. The relation between the ratio $\frac{V_a}{V_d}$ and the ratio $\frac{V_b}{V_c}$ is:View Solution
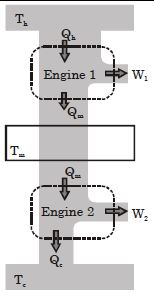
- 7Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\in_1$ and $\in_2$, respectively. The net efficiency of the combination is given by :View Solution
- 8A diatomic gas initially at $18^o C$ is compressed adiabatically to one-eighth of its original volume. The temperature after compression will beView Solution
- 9An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
- 10A carnot engine is made to work between $200\,^oC$ and $0\,^oC$ first and then between $0\,^oC$ and $-200\,^oC$. The ratio of efficiencies $\left( {\frac{{{\eta _2}}}{{{\eta _1}}}} \right)$ of the engine in two cases isView Solution
