A Container having $1\ mole$ of a gas at a temperature $27\ ^oC$ has a movable piston which maintains at constant pressure in container of $1\ atm.$ The gas is compressed until temperature becomes $127^oC.$ The work done is ........ $J$ $(C_p\ for\ gas\ is\ 7.03\ cal/mol-K)$
Medium
At constant pressure $W=P \Delta V=\mu R \Delta T$
$=1 \times 8.31 \times 100=831$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
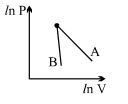
- 1The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
- 2A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
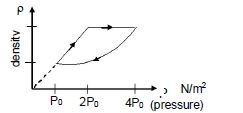
- 3An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 4Graph $A-B$ is an adiabatic curve. Choose the correct statementView Solution
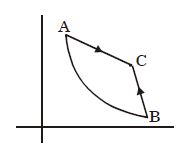
- 5In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
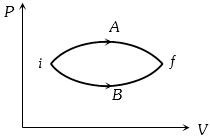
- 6View SolutionThe heat absorbed by a system in going through the given cyclic process is:
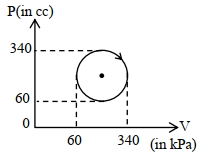
- 7View SolutionAn ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
- 8View SolutionThe slopes of isothermal and adiabatic curves are related as
- 9A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphereView Solution
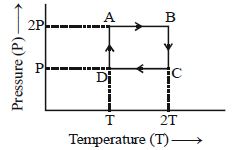
- 10One mole of an ideal gas having initial volume $V$, pressure $2P$ and temperature $T$ undergoes a cyclic process $ABCDA$ as shown below : The net work done in the complete cycle isView Solution