One mole of an ideal gas is taken from a to $b$ along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is $\mathrm{w}_{\mathrm{s}}$ and that along the dotted line path is $w_d$, then the integer closest to the ratio $w_d / w_5$ is
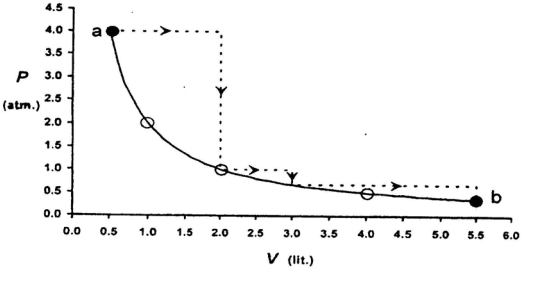

IIT 2010, Advanced
$w_d=4 \times 1.5+1 \times 1+2.5 \times 2 / 3=8.65$
Process is isothermal
$ \mathrm{w}_{\mathrm{s}}=2 \times 2.303 \log \frac{5.5}{0.5}=2 \times 2.303 \times \log 11=2 \times 2.303 \times 1.0414=4.79 $
$ \frac{\mathrm{w}_{\mathrm{d}}}{\mathrm{w}_{\mathrm{s}}}=\frac{8.65}{4.79}=1.80=2$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
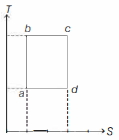
- 1An ideal gas is taken reversibly around the cycle $a-b-c-d-a$ as shown on the temperature $T$ - entropy $S$ diagram. The most appropriate representation of above cycle on a internal energy $U$ - volume $V$ diagram isView Solution
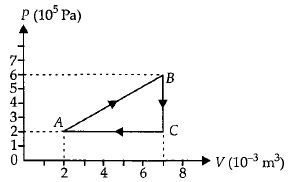
- 2A thermodynamic process is the pressure and volumes corresponding to some points in the figure are, $P_A = 3 \times 10^4 Pa$, $V_A = 2 \times 10^{-3}\, m^3$, $P_B = 8 \times 10^4 Pa$, $V_D = 5 \times 10^{-3}\,m^3$. In process $AB, 600\, J$ of heat and in process $BC, 200\, J$ of heat is added to the system. The change in the internal energy in process $AC$ would be .... $J$View Solution
- 3If $\Delta U$ and $\Delta W$ represent the increase in internal energy and work done by the system respectively in a thermodynamical process, which of the following is true?View Solution
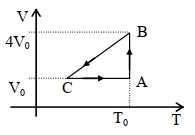
- 4One mole of an ideal gas in initial state $\mathrm{A}$ undergoes a cyclic process $A B C A$, as shown in the figure. Its pressure at $A$ is $\mathrm{P}_0$. Choose the correct option$(s)$ from the followingView Solution
$(A)$ Internal energies at $\mathrm{A}$ and $\mathrm{B}$ are the same
$(B)$ Work done by the gas in process $\mathrm{AB}$ is $\mathrm{P}_0 \mathrm{~V}_0 \ln 4$
$(C)$ Pressure at $C$ is $\frac{P_0}{4}$
$(D)$ Temperature at $\mathrm{C}$ is $\frac{\mathrm{T}_0}{4}$
- 5View SolutionThe work done in which of the following processes is equal to the internal energy of the system?
- 6An electric heater supplies heat to a system at a rate of $100\;W.$ If system performs work at a rate of $75$ Joules per second. At what rate (in $J/s$) is the internal energy increasing?View Solution
- 7View SolutionWhen you make ice cubes, the entropy of water
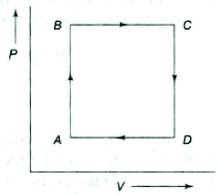
- 8A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
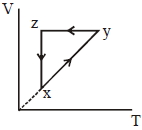
- 9A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
- 10A Carnot engine takes $3 \times {10^6}\,cal$. of heat from a reservoir at $627°C$, and gives it to a sink at $27°C.$ The work done by the engine isView Solution
