When you make ice cubes, the entropy of water
AIIMS 2006, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas at initial temperature $T$, undergoes a quasi-static process during which the volume $V$ is doubled. During the process, the internal energy $U$ obeys the equation $U=a V^3$, where $a$ is a constant. The work done during this process isView Solution
- 2A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
- 3An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine isView Solution
- 4The pressure and volume of an ideal gas are related as $\mathrm{PV}^{3 / 2}=\mathrm{K}$ (Constant). The work done when the gas is taken from state $A\left(P_1, V_1, T_1\right)$ to state $\mathrm{B}\left(\mathrm{P}_2, \mathrm{~V}_2, \mathrm{~T}_2\right)$ is :View Solution
- 5The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution
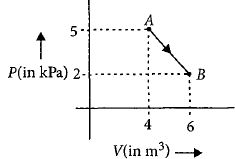
- 6One mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure.View Solution
The change in internal energy of the gas during the transition is ............$\;kJ$
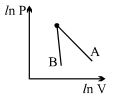
- 7The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
- 8A heat engine operates with the cold reservoir at temperature $324 K$. The minimum temperature of the hot reservoir, if the heat engine takes $300 \; J$ heat from the hot reservoir and delivers $180 \; J$ heat to the cold reservoir per cycle, is $\dots \; K .$View Solution
- 9View SolutionThe slopes of isothermal and adiabatic curves are related as
- 10View SolutionWhich statement is incorrect?
