One mole of ideal gas goes through process $P= 2V^2/(1+V^2)\,Pa$ then change in temperature of gas when volume changes from $V= 1\,m^3$ to $2\,m^3$ is
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The ratio of total energy of all molecules of one mole $O_2$ to the total energy of all molecules of two moles of $He$ at the same temprature isView Solution
- 2View SolutionThe average kinetic energy of a gas molecule can be determined by knowing
- 3One mole of an ideal gas $\left( {\frac{{{C_P}}}{{{C_V}}}\, = \gamma } \right)$ heated by law $P=\alpha V$ where $P$ is pressure of gas, $V$ is volume, $\alpha$ is a constant what is the heat capacity of gas in the process-View Solution
- 4A $25\times10^{-3}\, m^3$ volume cylinder is filled with $1\, mol$ of $O_2$ gas at room temperature $(300\, K)$. The molecular diameter of $O_2$, and its root mean square speed, are found to be $0.3\, nm$ and $200\, m/s$, respectively. What is the average collision rate (per second) for an $O_2$ molecule?View Solution
- 5At room temperature a diatomic gas is found to have an $r.m.s.$ speed of $1930\,ms^{-1}$. The gas isView Solution
- 6View SolutionAccording to kinetic theory of gases,
- 7A perfect gas at $27\,^oC$ is heated at constant pressure so as to double its volume. The final temperature of the gas will be, close to ...... $^oC$View Solution
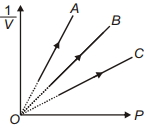
- 8Figure shows the isotherms of a fixed mass of an ideal gas at three temperatures $T_A, T_B$ and $T_C$, thenView Solution
- 9When pressure remaining constant, at what temperature will the $r.m.s.$ speed of a gas molecules increase by $10 \%$ of the r.m.s. speed at $STP$?View Solution
- 10A cylinder containing gas at $27\,^oC$ is divided into two parts of equal volume each $100\,^oc$ and at equal pressure by a piston of cross sectional area $10.85\,\, cm^2$. The gas in one part is raised in temperature to $100\,^oC$ while the other maintained at original temperature. The piston and wall are perfect insulators. How far will the piston move during the change in temperature.... $cm$ .View Solution
