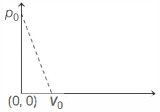
One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ is

KVPY 2015, Diffcult
(c)
Process given is
To find process equation, we use two point form of equation of straight line,
$y-y_1=\frac{y_2-y_1}{x_2-x_1}(x-x_1)$
Here, $(x, y,)=(0, p_0$, and $\left(x_2, y_2\right)=\left(V_0, 0\right)$ Process equation is
$p=p_0-\frac{p_0}{V_0} \cdot V$
As, $\quad p=\frac{R T}{V}$
$\Rightarrow \frac{R T}{V}=p_0-\frac{p_0}{V_0} \cdot V$
$\Rightarrow T=\frac{p_0 V}{R}\left(1-\frac{V}{V_0}\right)$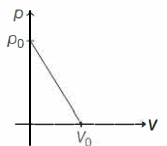
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process isView Solution
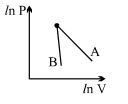
- 2The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
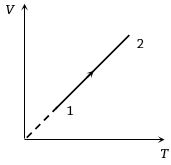
- 3Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
- 4A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 5A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close toView Solution
- 6Two Carnot engines $A$ and $B$ are operated in series. Engine $A$ receives heat from a reservoir at $600\,K$ and rejects heat to a reservoir at temperature $T$. Engine $B$ receives; heat rejected by engine $A$ and in turn rejects it to a reservoir at $100\,K$. If the efficiencies of the two engines $A$ and $B$ are represented by ${\eta _A}$ and ${\eta _B}$ respectively, then what is the value of $\frac{{{\eta _A}}}{{{\eta _B}}}$View Solution
- 7View SolutionIn which thermodynamic process, volume remains same
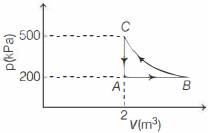
- 8A constant amount of an ideal gas undergoes the cyclic process $A B C A$ in the $p-V$ graph shown below. The path $B C$ is an isothermal. The work done by the gas during one complete cycle, beginning and ending at $A$ is nearly .......... $\,kJ$View Solution
- 9A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
- 10A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution