An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process is
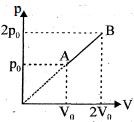

Medium
$\mathrm{C}=\mathrm{C}_{\mathrm{v}}+\frac{\mathrm{R}}{1-\mathrm{n}}$
in the given process
$\mathrm{P} \alpha \mathrm{V}$
$\mathrm{PV}^{-1}=\mathrm{const}$
$n=-1$
$\Rightarrow \mathrm{C}=\frac{3}{2} \mathrm{R}+\frac{\mathrm{R}}{1-(-1)}=2 \mathrm{R}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
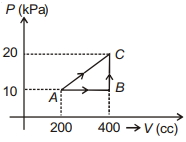
- 1If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
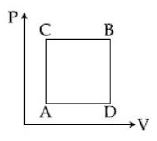
- 2A gas can be taken from $A$ to $B$ via two different processes $ACB$ and $ADB$. When path $ACB$ is used $60\, J$ of heat flows into the system and $30\, J$ of work is done by the system. If path $ADB$ is used work down by the system is $10\, J$. the heat flow into the system in path $ADB$ is ..... $J$View Solution
- 3Given that $1\,g$ of water in liquid phase has volume $1\,cm^3$ and in vapour phase $1671\, cm^3$ at atmospheric pressure and the latent heat of vaporization of water is $2256\,J/g;$ the change in the internal energy in joules for $1\,g$ of water at $373\,K$ when it changes from liquid phase to vapour phase at the same temperature is ....... $J$View Solution
- 4Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 5The temperature of food material in refrigerator is $4^{\circ} C$ and temperature of environment is $15^{\circ} C$. If carnot cycle is used in its working gas, then find its carnot efficiency.View Solution
- 6One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
- 7Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
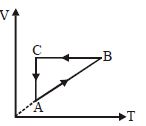
- 8A cyclic process $ABCA$ is shown in the $V-T$ diagram. Process on the $P-V$ diagram isView Solution
- 9View SolutionThe first operation involved in a Carnot cycle is
- 10The pressure and density of a diatomic gas $(\gamma = 7/5)$ change adiabatically from $(P, d)$ to $(P', d')$. If $\frac{{d'}}{d} = 32$, then $\frac{{P'}}{P}$ should beView Solution
