Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ is
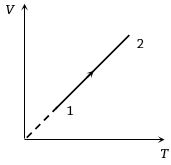

Medium
(b) $V-T$ graph is a straight line passing through origin. Hence, $V \propto T$ or $P = {\rm{constant}}$
$\therefore $ $\Delta Q = n{C_P}\Delta T$ and $\Delta U = n{C_V}\Delta T$
Also $\Delta W = \Delta Q - \Delta U = \mu \,({C_P} - {C_V})\,\Delta T$
$\therefore \,\,\,\,\,\,\frac{{\Delta Q}}{{\Delta W}} = \frac{{n{C_P}\Delta T}}{{n\,({C_P} - {C_V})\,\Delta T}}$$ = \frac{{{C_P}}}{{{C_P} - {C_V}}} = \frac{1}{{1 - \frac{{{C_V}}}{{{C_P}}}}}$
$\frac{{{C_V}}}{{{C_P}}} = \frac{3}{5}$ for helium gas. Hence $\frac{{\Delta Q}}{{\Delta W}} = \frac{1}{{1 - 3/5}} = \frac{5}{2}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes. - 2The specific heat capacity of a metal at low temperature $(T)$ is given as $C_p=32\left(\frac{ T }{400}\right)^{3}\;kJ\,k ^{-1}\, kg ^{-1}$. A $100\; g$ vessel of this metal is to be cooled from $20 \;K$ to $4\; K$ by a special refrigerator operating at room temperature $27^\circ c$). The amount of work required to cool the vessel isView Solution
- 3Slope of isotherm for a gas (having $\gamma=\frac{5}{3}$ ) is $3 \times 10^5 \,N / m ^2$. If the same gas is undergoing adiabatic change then adiabatic elasticity at that instant is ........... $\times 10^5 N / m ^2$View Solution
- 4View SolutionThe heat absorbed by a system in going through the given cyclic process is:
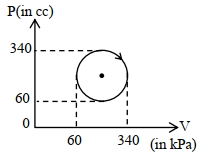
- 5A household refrigerator with a coefficient of performance $1.2$ removes heat from the refrigerated space at the rate of $60\ kJ/min$ .What would be cost of running this fridge for one month $\mathrm{Rs.}$ ..................... $(30\ days)$ (assuming each day it is used for $4$ hours and cost of one electrical unit is $6$ Rs.)View Solution
- 6View SolutionOut of the following which quantity does not depend on path
- 7The efficiency of carnot engine is $50\%$ and temperature of sink is $500\,K$ . If temperature of source is kept constant and its efficiency raised to $60\%$ , then the required temperature of the sink will be .... $K$View Solution
- 8$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 9An ideal gas is taken reversibly around the cycle $a-b-c-d-a$ as shown on the temperature $T$ - entropy $S$ diagram. The most appropriate representation of above cycle on a internal energy $U$ - volume $V$ diagram isView Solution
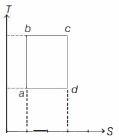
- 10When an ideal gas $(\gamma = 5/3$) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external workView Solution
