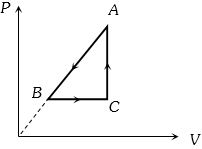
Therefore, temperature and hence, internal energy of the gas will decrease ($T \propto PV$) or $\Delta {U_{A \to B}} = $negative.
Further $\Delta {W_{A \to B}}$ is also negative as the volume of the gas is decreasing. Thus $\Delta {Q_{A \to B}}$ is negative.
In process $B$ to $C$ pressure of the gas is constant while volume is increasing.
Hence temperature should increase or $\Delta {U_{B \to C}}$= positive.
During $C$ to $A$ volume is constant while pressure is increasing.
Therefore, temperature and hence, internal energy of the gas should increase or $\Delta {U_{C \to A}}$= positive.
During process $CAB$ volume of the gas is decreasing. Hence, work done by the gas is negative.
Download our appand get started for free
Similar Questions
- 1One mole of a monatomic ideal gas is taken through a cycle $ABCDA$ as shown in the $P-V$ diagram. Column $II$ gives the characteristics involved in the cycle. Match them with each of the processes qiven in Column $I$View Solution
Column $I$ Column $II$ $(A)$ Process $A \rightarrow B$ $(p)$ Internal energy decreases. $(B)$ Process $B \rightarrow C$ $(q)$ Internal energy increases. $(C)$ Process $C \rightarrow D$ $(r)$ Heat is lost. $(D)$ Process $D \rightarrow A$ $(s)$ Heat is gained. $(t)$ Work is done on the gas. 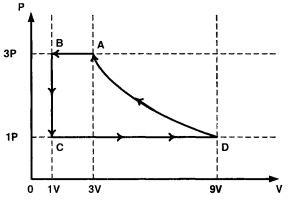
- 2View SolutionWhich of the following is not thermodynamical function
- 3$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
- 4$P_i, V_i$ and $P_f$, $V_f$ are initial and final pressures and volumes of a gas in a thermodynamic process respectively. If $PV^n =$ constant, then the amount of work done isView Solution
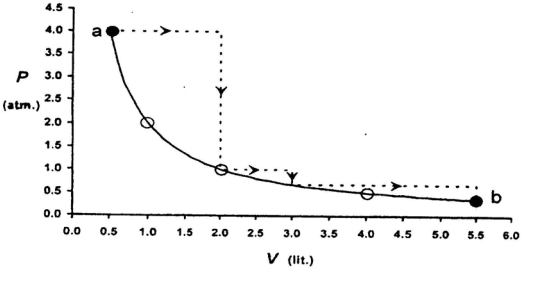
- 5One mole of an ideal gas is taken from a to $b$ along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is $\mathrm{w}_{\mathrm{s}}$ and that along the dotted line path is $w_d$, then the integer closest to the ratio $w_d / w_5$ isView Solution
- 6View SolutionFor a reversible process, necessary condition is
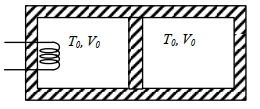
- 7A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as shown in the figure below. On each side of the partition, there is one mole of an ideal gas, with specific heat at constant volume, $C_v=2 R$. Here, $R$ is the gas constant. Initially, each side has a volume $V_0$ and temperature $T_0$. The left side has an electric heater, which is turned on at very low power to transfer heat $Q$ to the gas on the left side. As a result the partition moves slowly towards the right reducing the right side volume to $V_0 / 2$. Consequently, the gas temperatures on the left and the right sides become $T_L$ and $T_R$, respectively. Ignore the changes in the temperatures of the cylinder, heater and the partition.View Solution
($1$) The value of $\frac{T_R}{T_0}$ is
$(A)$ $\sqrt{2}$ $(B)$ $\sqrt{3}$ $(C)$ $2$ $(D)$ $3$
($2$) The value of $\frac{Q}{R T_0}$ is
$(A)$ $4(2 \sqrt{2}+1)$ $(B)$ $4(2 \sqrt{2}-1)$ $(C)$ $(5 \sqrt{2}+1)$ $(D)$ $(5 \sqrt{2}-1)$
Give the answer or qution ($1$) and ($2$)
- 8View SolutionWhich one is the correct option for the two different thermodynamic processes ?
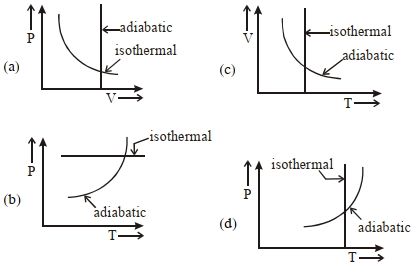
- 9For $P-V$ diagram of a thermodynamic cycle as shown in figure, process $B C$ and $D A$ are isothermal. Which of the corresponding graphs is correct?View Solution
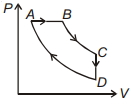
- 10When $1\, gm$ of water at ${0^o}C$ and $1 \times {10^5}\;N/{m^2}$ pressure is converted into ice of volume $1.091\;c{m^2}$, the external work done will beView Solution
