Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The pressure of a gas changes linearly with volume from $A$ to $B$ as shown in figure. If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be $............\,J$View Solution
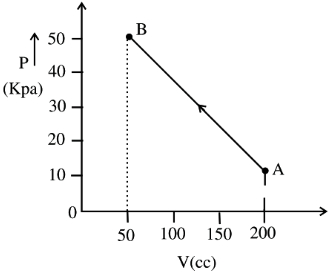
- 2An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
- 3View SolutionWhich of the following can be coefficient of performance of refrigerator?
- 4In an adiabatic process where in pressure is increased by $\frac{2}{3}\% $ if $\frac{{{C_p}}}{{{C_v}}} = \frac{3}{2},$ then the volume decreases by aboutView Solution
- 5An insulator container contains $4\, moles$ of an ideal diatomic gas at temperature $T.$ Heat $Q$ is supplied to this gas, due to which $2 \,moles$ of the gas are dissociated into atoms but temperature of the gas remains constant. ThenView Solution
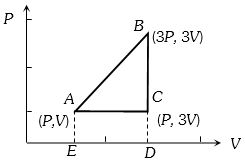
- 6A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
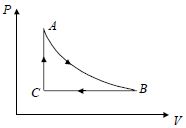
- 7Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$View Solution
- 8View SolutionThe first law of thermodynamics is concerned with the conservation of
- 9In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
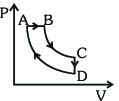
- 10A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution