Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe order of boiling point of primary (1), secondary (2) and tertiary (3) alcohols is:
- 2View SolutionWhich of the following alcohols is the least soluble in water?
- 3View SolutionWhich of the following statement is not correct?
- 4View SolutionWhat type of spectroscopy would be the best tool to analyze a clear and colorless solution for the presence of a functional group like an alcohol group (−OH)?
- 5View SolutionMaximum boiling point would be shown by:
- 6View Solution
- 7View SolutionWhich is the best reagent to convert cyclohexanol into cyclohexene?
- 8View SolutionHeating together of sodium ethoxide and ethyl iodide will give:
- 9Methanol can be prepared when$:$View Solution
- 10View SolutionThe IUPAC name is:
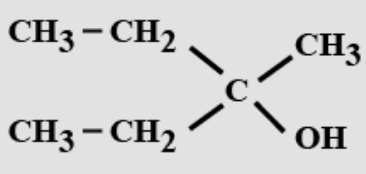

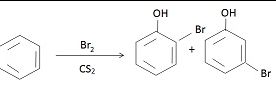
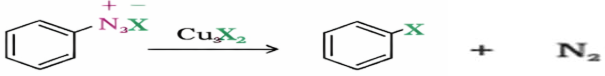 is-
is-