પક્રિયા $2 NO + Br _2 \rightarrow 2 NOBr$
નીચે આપેલ પ્રક્કિયાવિધી દ્વારા થઈ રહી છે.
$NO + Br _2 \Leftrightarrow NOBr _2 \text { (fast) }$
$NOBr _2+ NO \rightarrow 2 NOBr$(ધીમી)
પ્રક્રિયાનો સમગ્ર ક્રમ $........$
JEE MAIN 2023, Medium
b
$RDS : NOBr _2+ NO \rightarrow 2 NOBr$
$RDS : NOBr _2+ NO \rightarrow 2 NOBr$
$\left. r = K \left[ NOBr _2\right] NO \right] \quad--- \text { (i) }$
$Keq =\frac{\left[ NOBr _2\right]}{[ NO ]\left[ Br _2\right]}----- \text { (ii) }$
$\text { From (i) and (ii) }$
$r = K \cdot Keq \cdot[ NO ]\left[ Br _2\right][ NO ]$
$r = K ^{\prime}[ NO ]^2\left[ Br _2\right]$
$\text { Overall order }=3$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View Solutionપ્રથમ ક્રમની પ્રક્રિયાનો વિશિષ્ટ વેગ અચળાંક ............. પર આધાર રાખે છે.
- 2પદાર્થ $A$ નું દ્રાવણમાં વિઘટન પ્રથમ ક્રમની ગતિકીને અનુસરે છે. પાત્ર $-I$ એ $1\, MA$ નુ $1$ લિટર દ્રાવણ ધરાવે છે. પાત્ર $-II\, 0.6\, MA$ $100\, mL$ દ્રાવણ ધરાવે છે. $8$ કલાકમાં પાત્ર $-I$ માંના $A$ ની સાંદ્રતા $0.25\, M$ થાય, તો પાત્ર $- II$ માંના $A$ ની સાંદ્રતા $0.3$ થવા ......... $hr$ લાગશે.View Solution
- 3જો પ્રક્રિયાનો $t_{1/2} = 69.3$ સેકન્ડ છે અને દર અચળાંક $10^{-2}$ પ્રતિ સેકન્ડ છે તો પ્રક્રિયા ક્રમ.......View Solution
- 4એક વાયરૂપ પ્રક્રિયાનો વેગ $r = K\,[x]\, [y]$ છે. જો એકાએક પાત્રનુ કદ ઘટાડીને શરૂઆતના કદથી $1/4$ જેટલુ કરવામાં આવે તો પ્રક્યિાનો વેગ ............View Solution
- 5પ્રક્રિયકો $A$ અને $B$ ને સમાવતી પ્રક્રિયાનો વેગ = $= k[A ]^n[B]^m$ છે. જો A ની સાંદ્રતા બમણી અને B ની સાંદ્રતા અડધી કરીએ તો તવા વેગ અને મૂળ વગનો ગુણોત્તર ......... થશે.View Solution
- 6એક નિશ્ચિત રાસાયણિક પ્રક્રિયા $X \rightarrow Y$ માટે, નીપજના બનવાનો દર વિરુદ્ધ સમયનો ગ્રાફ (આલેખ) એ આકૃતિમાં દર્શાવેલ છે. નીચે આપેલામાંથી સાચાં વિધાન/નોની સંખ્યા $..............$ છે.View Solution
$(A)$ સમગ્ર પ્રક્રિયાનો ક્રમ છે.
$(B)$ આ પ્રક્રિયાનો ક્રમ શોધી શકાતો નથી.
$(C)$ $I$ અને $III$ વિભાગ માં, પ્રક્રિયા અનુક્રમે પ્રથમ અને શૂન્ય ક્રમની છે.
$(D)$ વિભાગ $II$ માં, પ્રક્રિયા પ્રથમક્રમની છે.
$(E)$ વિભાગ $II$ માં, પ્રક્રિયાનો ક્રમ $0.1$ થી $0.9$ વિસ્તાર માં છે.
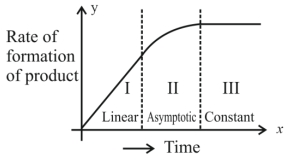
- 7View Solutionજો સાંદ્રતા મોલ/લિટરમાં દર્શાવવામાં આવે તો પ્રથમક્રમની પ્રક્રિયા માટે વેગ અચળાંકનો એકમ........ છે.
- 8$A + 2B\rightarrow $ નિપજ $ (P)$ પ્રક્રિયાનો દર નિયમ $\frac{{d[P]}}{{dt}}\,\, = \,\,K{[A]^2}[B]$ છે. જો મોટા પ્રમાણમાં $ [A]$ લેવામાં આવે તો પ્રક્રિયાનો ક્રમ શું થશે?View Solution
- 9પ્રથમ ક્રમની પ્રક્રિયાનો અર્ધ આયુષ્ય સમય $1386\, s$ છે. તો પ્રક્રિયાનો વિશિષ્ટ વેગઅચળાંક ............ થશે.View Solution
- 10View Solutionનીચે પૈકી કયું વિધાન સાચું છે?
