At which temperature the r.m.s. velocity of a hydrogen molecule equal to that of an oxygen molecule at $47^{\circ} \mathrm{C}$ ?
JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn which of the following gas the root mean square velocity will be minimum (at const. temperature)
- 2A container is divided into two equal parts $I$ and $II$ by a partition with a small hole of diameter $d$. The two partitions are filled with same ideal gas, but held at temperatures $T_{ I }=150 \,K$ and $T_{ II }=300 \,K$ by connecting to heat reservoirs. Let $\lambda_{1}$ and $\lambda_{1 I}$ be the mean free paths of the gas particles in the two parts, such that $d >> \lambda_{ I }$ and $d >> \lambda_{ II }$. Then, the $\lambda_{ I } / \lambda_{ II }$ is close toView Solution
- 3Match List$-I$ with List$-II:$View Solution
List$-I$ List$-II$ $(A)$ $3$ Translational degrees of freedom $(I)$ Monoatomic gases $(B)$ $3$ Translational,$2$ rotational degrees of freedoms $(II)$ Polyatomic gases $(C)$ $3$ Translational,$2$ rotational and $1$ vibrational degrees of freedom $(III)$ Rigid diatomic gases $(D)$ $3$ Translational,$3$ rotational and more than one vibrational degrees of freedom $(IV)$ Nonrigid diatomic gases Choose the correct answer from the options given below:
- 4The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
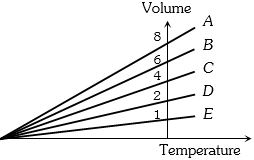
- 5A gaseous mixture consists of $16\,g$ of helium and $16\,g$ of oxygen. The ratio $\frac{{{C_P}}}{{{C_V}}}$ of the mixture isView Solution
- 6At $0 \;K$ which of the following properties of a gas will be zeroView Solution
- 7View SolutionMaxwell distribution function is shown in figure for different gases, which of the following is correct matching?
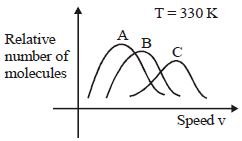
- 8Which one of the following schematic graphs best represents the variation of $p V$ (in Joules) versus $T$ (in Kelvin) of one mole of an ideal gas? (The dotted line represents $p V=T$ )View Solution
- 9$2\, gm$ of ${O_2}$ gas is taken at $27°C$ and pressure $76\, cm.$ Of $Hg$. Then find out volume of gas (in litre)View Solution
- 10At room temperature a diatomic gas is found to have an $r.m.s.$ speed of $1930\,ms^{-1}$. The gas isView Solution
