પ્રક્રિયા $NH_4^+ + NO_2^- \to N_2 + 2H_2O$ માટે પ્રાયોગિક માહિતી નીચે મુજબ છે. તો પ્રકિયા માટે વેગનિયમ જણાવો.
| No | $[NH_4^+]$ | $[NO_2^-]$ | rate of reaction |
| $1.$ | $0.24\, M$ | $0.10\, M$ | $7.2 \times {10^{ - 6}}$ |
| $2.$ | $0.12\, M$ | $0.10\, M$ | $3.6 \times {10^{ - 6}}$ |
| $3.$ | $0.12\, M$ | $0.15\, M$ | $5.4 \times {10^{ - 6}}$ |
Medium
b
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1જો સાંદ્રતા એકમ $n$ ગણો ઘટે છે તો પ્રથમ ક્રમના દર અચળાંકનું મૂલ્ય શું થશે?View Solution
- 2સામાન્ય રીતે, પાછળની અને આગળની પ્રતિક્રિયાઓ માટે, સક્રિયકરણ શક્તિઓ ${E_b}$ અને ${E_f}$ સાથે, ઉષ્માશોષક પ્રક્રિયા $X$ થી $Y$ ધ્યાનમાં લોView Solution
- 3જો પ્રક્રિયક $ B$ ની સાંદ્રતા બમણી થાય તો પ્રક્રિયક $A$ અને $B$ વચ્ચેની પ્રક્રિયાનો દર પ્રારંભિક દર $1/4$ જેટલો થાય છે. પ્રક્રિયક $B$ ના સંદર્ભમાં પ્રક્રિયાનો ક્રમ ...... થશે.View Solution
- 4$A + B \rightarrow $ નિપજ પ્રક્રિયા માટેનો દર નિયમ દર $= K[A]^1[B]^2$ છે. તો નીચેનામાંથી કયું વિધાન ખોટું છે?View Solution
- 5$H_2$ ની $I_2$ સાથેની પ્રક્રિયા માટે, $327\,^oC$ પર વેગ અચળાંક $2.5\times 10^{-4}\,dm^3\,mol^{-1}\,s^{-1}$ અને $527\,^oC$ પર $1.0\,dm^3\,mol^{-1}\,s^{-1}$ છે. તો પ્રક્રિયાતી સક્રિયકરણ ઊર્જા ($kJ\,mol^{-1}$ માં) જણાવો. $(R = 8.314\,J\,K^{-1}\,mol^{-1} )$View Solution
- 6$2A + B \rightarrow C$ પ્રક્રિયા માટેનું દર સમીકરણ : દર $= k[A][B]$ મળે છે. તો આ પ્રક્રિયાનો સંબંધ માટે સાચું વિધાન કયુુ છે?View Solution
- 7પ્રથમક્રમની પ્રક્રિયા માટે શરૂઆતની સાંદ્રતામાં $1/4$ જેટલો ઘટાડો થવા માટે લાગતો સમય $20$ મિનિટ છે. તો શરૂઆતની સાંદ્રતા માં $1/16 $ જેટલો ઘટાડો થવા માટે લાગતો સમય......... $\min.$ હશે.View Solution
- 8આપેલ આલેખ પ્રક્રિયાની ગતિકી દર્શાવે છે.View Solution
શૂન્ય અને પ્રથમક્રમ પ્રક્રિયા માટે $y$ અને $x$ અક્ષો અનુક્રમે...
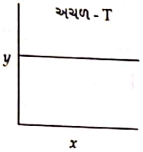
- 9પ્રતિવર્તી પ્રકિયા $ A \rightleftharpoons B $ માટે પ્રકિયક અને નીપજના અર્ધઆયુષ્ય સમય અનુકમે $15\, s$ અને $18\,s$ છે. જો બંને પ્રક્રિયાઓ પ્રથમ કમની ગતિકીને અનુસરતી હોય, તો પ્રક્રિયાનો સંતુલન અચળાંક $(K_c )$ કેટલો થશે ?View Solution
- 10View Solutionપ્રથમ ક્રમ પ્રક્રિયાનું સાચુ વિધાન ....... થશે.
