Read the given statements and decide which is/are correct on the basis of kinetic theory of gases
$(I)$ Energy of one molecule at absolute temperature is zero
$(II)$ $r .m.s.$ speeds of different gases are same at same temperature
$(III)$ For one gram of all ideal gas kinetic energy is same at same temperature
$(IV)$ For one mole of all ideal gases mean kinetic energy is same at same temperature
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A very tall vertical cylinder is filled with a gas of molar mass $M$ under isothermal conditions at temperature $T.$ The density and pressure of the gas at the base of the container is $\rho_0$ and $p_0$, respectively Choose the correct statement(s)View Solution
- 2The average degree of freedom per molecule of a gas is $6$. The gas performs $25\ J$ work, while expanding at constant pressure. The heat absorbed by the gas is .... $J$View Solution
- 3Pressure $Vs$ temperature graph of an ideal gas is shown in figure. Density of the gas at point $A$ is $\rho _0$ , Density at $B$ will beView Solution
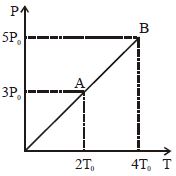
- 4One mole of an ideal monoatomic gas at temperature $T_0$ expands slowly according to the law $P/V$ = constant. If the final temperature is $2 \,\,T_0$, heat supplied to the gas is :View Solution
- 5View SolutionKinetic theory of gases provide a base for
- 6The average distance between molecules of an ideal gas at $STP$ is approximately of the order ofView Solution
- 7View SolutionThe gas which obeys Boyle's law for maximum range of temperature is
- 8At $NTP,$ sample of equal volume of chlorine and oxygen is taken. Now ratio of No. of moleculesView Solution
- 9Open container contains $x$ mole of gas at $T$ $kelvin$ If temperature is tripled, then the amount of gas exit the container isView Solution
- 10A soap bubble of radius $r$ has monoatomic ideal- gas inside. The gas is heated in such a manner that bubble remains in mechanical equillibrium. Assuming that the soap material of the bubble has no heat capacity, the molar heat capacity of the gas in the process will beView Solution
(Neglect atmospheric pressure)
