The gas which obeys Boyle's law for maximum range of temperature is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At a given temperature if ${V_{rms}}$ is the root mean square velocity of the molecules of a gas and ${V_s}$ the velocity of sound in it, then these are related as $\left( {\gamma = \frac{{{C_P}}}{{{C_v}}}} \right)$View Solution
- 2A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300 K$. The ratio of the average rotational kinetic energy per ${O_2}$ molecule to that per ${N_2}$ molecule isView Solution
- 3A container with insulating walls is divided into two equal parts by a partition fitted with a valve. One part is filled with an ideal gas at a pressure $P$ and temperature $T$, whereas the other part is completely evacuated. If the valve is suddenly opened, the pressure and temperature of the gas will beView Solution
- 4View SolutionIt is possible for a substance to coexist in all three phases in equilibrium, when the substance is at
- 5A cylinder of fixed capacity of $44.8 \,litres$ contains helium gas at standard temperature and pressure. The amount of heat needed to raise the temperature of gas in the cylinder by $20.0^{\circ} C$ will be .............. $J$(Given gas constant $R =8.3 \,JK ^{-1}- moI ^{-1}$ )View Solution
- 6A cylindrical tube of cross-sectional area $A$ has two air tight frictionless pistons at its two ends. The pistons are tied with a straight two ends. The pistons are tied with a straight piece of metallic wire. The tube contains a gas at atmospheric pressure $P_0$ and temperature $T_0$. If temperature of the gas is doubled then the tension in the wire isView Solution
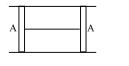
- 7A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $1.4$. Vessel is moving with speed $v$ and is suddenly brought to rest. Assuming no heat is lost to the surrounding and vessel temperature of the gas increases by ... ( $R =$ universal gas constant )View Solution
- 8A cylinder containing gas at $27\,^oC$ is divided into two parts of equal volume each $100\,^oc$ and at equal pressure by a piston of cross sectional area $10.85\,\, cm^2$. The gas in one part is raised in temperature to $100\,^oC$ while the other maintained at original temperature. The piston and wall are perfect insulators. How far will the piston move during the change in temperature.... $cm$ .View Solution
- 9Select the incorrect statement$(s)$View Solution
- 10Three particles have speeds of $2u$ , $10u$ and $11u$. Which of the following statements is correct?View Solution
