We sit in the room with windows open. Then,
KVPY 2011, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$2$ mole of $N_2$ gas at $27\,^oC$ is mixed with $1$ mole $He$ gas at $-73\,^oC$. Find $T_{mix}$ of the mixtureView Solution
- 2View SolutionFor an ideal gas of diatomic molecules
- 3The temperature of an ideal gas is reduced from $927^\circ C$ to $27^\circ C$. The $r.m.s.$ velocity of the molecules becomesView Solution
- 4View SolutionThe effect of temperature on Maxwell's speed distribution is correctly shown by
- 5The average translational kinetic energy of a hydrogen gas molecules at $NTP$ will beView Solution
[Boltzmann’s constant ${k_B} = 1.38 \times {10^{ - 23}}J/K]$
- 6$40$ calories of heat is needed to raise the temperature of $1\, mole$ of an ideal monoatomic gas from $20°C$ to $30°C$ at a constant pressure. The amount of heat required to raise its temperature over the same interval at a constant volume $(R = 2\,calorie\,mol{e^{ - 1}}{K^{ - 1}})$ is ..... $calorie$View Solution
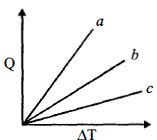
- 7Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond toView Solution
- 8The density of a gas at normal pressure and $27°C$ temperature is $24.$ Keeping the pressure constant, the density at $127°C$ will beView Solution
- 9At constant pressure, the ratio of increase in volume of an ideal gas per degree raise in kelvin temperature to it's original volume is $(T =$ absolute temperature of the gas$)$View Solution
- 10Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ If the internal energy of He sample of $100\,\,J$ and that of the hydrogen sample is $200\,\,J$, then the internal energy of the mixture is ..... $J$View Solution
