Under which of the following conditions is the law $PV = RT$ obeyed most closely by a real gas
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$N\,moles$ of a diatomic gas in a cylinder are at a temperature $T$. Heat is supplied to the cylinder such that the temperature remains constant but $n\,moles$ of the diatomic gas get converted into monoatomic gas. What is the change in the total kinetic energy of the gas ?View Solution
- 2From the following $V-T$ diagram we can concludeView Solution
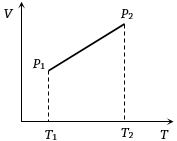
- 3View SolutionWhich of the following plots represents schematically the dependence of the time period of a pendulum, if measured and plotted as a function of the amplitude of its oscillations? (Note amplitude need not be small)
- 4View SolutionAt absolute zero temperature, pressure of a gas will be
- 5An ideal gas filled in a cylinder occupies volume $V$. The gas is compressed isothermally to the volume $V/3$. Now, the cylinder valve is opened and the gas is allowed to leak keeping temperature same. What percentage of the number of molecules should escape to bring the pressure in the cylinder back to its original value?View Solution
- 6A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.View Solution
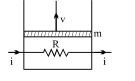
- 7View SolutionTwo vessels having equal volume contains molecular hydrogen at one atmosphere and helium at two atmospheres respectively. If both samples are at the same temperature, the mean velocity of hydrogen molecules is
- 8At what temperature will the oxygen molecules have the same root mean square speed as hydrogen molecules at $200 \,K$ ....... $K$View Solution
- 9The lowest pressure (the best Vacuum) that can be created in the laboratory at $27$ degree is $10^{-11} \;{mm}$ of $Hg$. At this pressure, the number of ideal gas molecules per ${cm}^{3}$ will beView Solution
- 10If mass of $He$ atom is $4$ times that of hydrogen atom then mean velocity of $He$ isView Solution
