Select the incorrect statement about the specific heats of a gaseous system.
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A monoatomic gas of mass $4.0\, u$ is kept in an insulated container. Container is moving with velocity $30 \,m / s$. If container is suddenly stopped then change in temperature of the gas $\left( R =\right.$ gas constant) is $\frac{ x }{3 R } .$ Value of $x$ is ..........View Solution
- 2A vessel contains $14\,g$ of nitrogen gas at a temperature of $27^{\circ}\,C$. The amount of heat to be transferred to the gap to double the r.m.s. speed of its molecules will be $......J$ $\left(\right.$ Take $R =8.32\,J\,mol ^{-1} k ^{-1}$ )View Solution
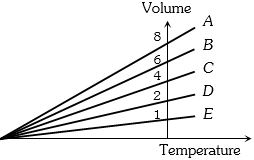
- 3The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
- 4The velocity of the molecules of a gas at temperature $120\,K$ is $v$. At ...... $K$ temperature will the velocity be $2v$View Solution
- 5At constant volume the specific heat of a gas is $\frac{{3R}}{2}$, then the value of $'\gamma '$ will be ....View Solution
- 6A system consists of two types of gas molecules $A$ and $B$ having same number density $2 \times$ $10^{25}\, / {m}^{3}$. The diameter of ${A}$ and ${B}$ are $10\, \stackrel{\circ}{{A}}$ and $5\, \stackrel{\circ}{{A}}$ respectively. They suffer collision at room temperature. The ratio of average distance covererd by the molecule $A$ to that of $B$ between two successive collision is $.....\,\times 10^{-2}$View Solution
- 7$N$ molecules each of mass $m$ of gas $A$ and $2N$ molecules each of mass $2m$ of gas $B$ are contained in the same vessel at temperature $T.$ The mean square of the velocity of molecules of gas $B$ is ${v^2}$ and the mean square of $x$ component of the velocity of molecules of gas $A$ is ${w^2}$. The ratio $\frac{{{w^2}}}{{{v^2}}}$ isView Solution
- 8A container is filled with $20$ moles of an ideal diatomic gas at absolute temperature $T$. When heat is supplied to gas temperature remains constant but $8$ moles dissociate into atoms. Heat energy given to gas is .........View Solution
- 9If the root mean square velocity of hydrogen molecule at a given temperature and pressure is $2 \mathrm{~km} / \mathrm{s}$, the root mean square velocity of oxygen at the same condition in $\mathrm{km} / \mathrm{s}$ is :View Solution
- 10If the pressure of an ideal gas contained in a closed vessel is increased by $0.5\%,$ the increase in temperature is $2K.$ The initial temperature of the gas is ...... $^oC$View Solution
