If the pressure of an ideal gas contained in a closed vessel is increased by $0.5\%,$ the increase in temperature is $2K.$ The initial temperature of the gas is ...... $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Ideal monoatomic gas is taken through a process $dQ = 2dU.$ The molar heat capacity for the processView Solution
- 2Calculate the value of mean free path $(\lambda)$ for oxygen molecules at temperature $27^{\circ}\, C$ and pressure $1.01 \times 10^{5} \,Pa$. Assume the molecular diameter $0.3 \,nm$ and the gas is ideal. $\left( k =1.38 \times 10^{-23}\, J\,K ^{-1}\right)$ (in $nm$)View Solution
- 3View SolutionThe ratio of mean kinetic energy of hydrogen and oxygen at a given temperature is
- 4A monoatomic gas of mass $4.0\, u$ is kept in an insulated container. Container is moving with velocity $30 \,m / s$. If container is suddenly stopped then change in temperature of the gas $\left( R =\right.$ gas constant) is $\frac{ x }{3 R } .$ Value of $x$ is ..........View Solution
- 5The mean free path of gas molecules depends on $(d =$ molecular diameter$)$View Solution
- 6$1\ mole$ of a gas with $\gamma$ = $7/5$ is mixed with $1\ mole$ of a gas with $\gamma$ = $5/3$, then the value of $\gamma$ for the resulting mixture isView Solution
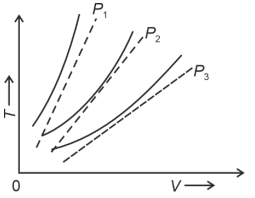
- 7The following graph represents the $T-V$ curves of an ideal gas (where $T$ is the temperature and $V$ the volume) at three pressures $P_1, P_2$ and $P_3$ compared with those of Charles's law represented as dotted lines.View Solution
Then the correct relation is:
- 8$0.056 \,kg$ of Nitrogen is enclosed in a vessel at a temperature of $127\,^{\circ} C$. The amount of heat required to double the speed of its molecules is k cal. (Take $R =2$ $cal \,mole$ $^{-1} K ^{-1}$ )View Solution
- 9One mole of ideal gas goes through process $P= 2V^2/(1+V^2)\,Pa$ then change in temperature of gas when volume changes from $V= 1\,m^3$ to $2\,m^3$ isView Solution
- 10The temperature of a gas having $2.0 \times 10^{25}$ molecules per cubic meter at $1.38 \mathrm{~atm}$ (Given, $\mathrm{k}=$ $\left.1.38 \times 10^{-23} \mathrm{JK}^{-1}\right)$ is :View Solution
