A monoatomic gas of mass $4.0\, u$ is kept in an insulated container. Container is moving with velocity $30 \,m / s$. If container is suddenly stopped then change in temperature of the gas $\left( R =\right.$ gas constant) is $\frac{ x }{3 R } .$ Value of $x$ is ..........
JEE MAIN 2021, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At temperature $T,$ the $r.m.s.$ speed of helium molecules is the same as $r.m.s.$ speed of hydrogen molecules at normal temperature and pressure. The value of $T$ is ....... $^oC$View Solution
- 2Relationship between $P,V,$ and $E$ for a gas isView Solution
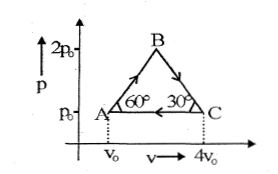
- 3Four moles of an ideal monoatomic gas undergoes a cyclic process $ABCA$ as shown in figure find ratio of temperature of $A$ and $B$ .View Solution
- 4Find $\gamma$ for the mixture of $11\, gm CO _{2}$ and $14\, gm N _{2} ?$View Solution
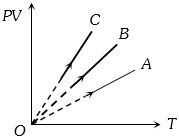
- 5$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
- 6View SolutionInternal energy of a non-ideal gas depends on ..........
- 7$Assertion :$ Air pressure in a car tyre increases during driving.View Solution
$Reason :$ Absolute zero temperature is not zero energy temperature. - 8The graph between pressure $(P)$ and $\frac{1}{V}$ ($V$ is volume) for constant temperature process in ideal equation $(PV = nRT)$View Solution
- 9A gas at $ 27\ ^oC$ has a volume $V$ and pressure $P$. On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ....... $^oC$View Solution
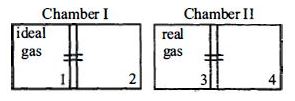
- 10There are two identical chambers, completely thermally insulated from surroundings. Both chambers have a partition wall dividing the chambers in two compartments. Compartment $1$ is filled with an ideal gas and Compartment $3$ is filled with a real gas. Compartments $2$ and $4$ are vacuum . A small hole (orifice) is made in the partition walls and the gases are allowed to expand in vacuumView Solution
Statement $-1$ : No change in the temperature of the gas takes place when ideal gas expands in vacuum. However, the temperature of real gas goes down (cooling) when it expands in vacuum
Statement $-2$ : The internal energy of an ideal gas is only kinetic. The internal energy of a real gas is kinetic as well as potential