A vessel contains $14\,g$ of nitrogen gas at a temperature of $27^{\circ}\,C$. The amount of heat to be transferred to the gap to double the r.m.s. speed of its molecules will be $......J$ $\left(\right.$ Take $R =8.32\,J\,mol ^{-1} k ^{-1}$ )
JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas has molecules with $5\,$ degrees of freedom. The ratio of specific heats at constant pressure $(C_p)$ and at constant volume $(C_v)$ isView Solution
- 2View SolutionA gas is enclosed in a closed pot. On keeping this pot in a train moving with high speed, the temperature of the gas
- 3$Assertion$: The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5\, times$ the product of its pressure and its volume.View Solution
$Reason$ : The molecules of a gas collide with each other and the velocities of the molecules change due to the collision. - 4If ${V_H},\,\,{V_N}$ and ${V_O}$ denote the root-mean square velocities of molecules of hydrogen, nitrogen and oxygen respectively at a given temperature, thenView Solution
- 5View SolutionSelect the incorrect statement about the specific heats of a gaseous system.
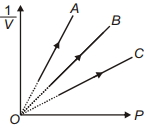
- 6Figure shows the isotherms of a fixed mass of an ideal gas at three temperatures $T_A, T_B$ and $T_C$, thenView Solution
- 7Consider an ideal gas confined in an isolated closed chamber. As the gas undergoes an adiabatic expansion, the average time of collision between molecules increases as $V ^q$, where $V$ is the volume of the gas. The value of $q$ is $\left( {\gamma = \frac{{{C_P}}}{{{C_V}}}} \right)$View Solution
- 8The temperature of argon, kept in a vessel, is raised by $1^\circ C$ at a constant volume. The total heat supplied to the gas is a combination of translational and rotational energies. Their respective shares areView Solution
- 9When a gas in a closed vessel was heated so as to increase its temperature by ${5^o}C$, there occurred an increase of $1\%$ in its pressure. The original temperature of the gas was ...... $^oC$View Solution
- 10View SolutionOn the basis of kinetic theory of gases, the gas exerts pressure because its molecules:
