Surface of the lake is at $2^{\circ} C$. The temperature of the bottom of the lake is ....... $^{\circ} C$
Easy
(c)
The densest layer of water will be at bottom. The density of water is maximum at $4^{\circ}\,C$. So the temperature of bottom of lake will be $4^{\circ}\,C$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe quantity of heat which crosses unit area of a metal plate during conduction depends upon
- 2View SolutionA thin paper cup filled with water does not catch fire when placed over a flame. This is because
- 3View SolutionWater and turpentine oil (specific heat less than that of water) are both heated to same temperature. Equal amounts of these placed in identical calorimeters are then left in air
- 4A black body is at a temperature of $5760\ K$. The energy of radiation emitted by the body at wavelength $250\ nm$ is $U_1$, at wavelength $500\ nm$ is $U_2$ and that at $1000\ nm$ is $U_3$. Wien's constant, $b = 2.88 \times 10^6\ nm\ K$. Which of the following is correct?View Solution
- 5One end of a copper rod of length $1.0\;m$ and area of cross-section ${10^{ - 3}}$ is immersed in boiling water and the other end in ice. If the coefficient of thermal conductivity of copper is $92\;cal/m{\rm{ - }}s{{\rm{ - }}^o}C$ and the latent heat of ice is $8 \times {10^4}cal/kg$, then the amount of ice which will melt in one minute isView Solution
- 6A large cylindrical rod of length $L$ is made by joining two identical rods of copper and steel of length $(\frac {L}{2})$ each . The rods are completely insulated from the surroundings. If the free end of copper rod is maintained at $100\,^oC$ and that of steel at $0\,^oC$ then the temperature of junction is........$^oC$ (Thermal conductivity of copper is $9\,times$ that of steel)View Solution
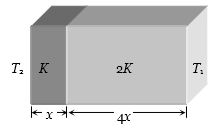
- 7The temperature of the two outer surfaces of a composite slab, consisting of two materials having coefficients of thermal conductivity $K$ and $2K$ and thickness $x$ and $4x$ , respectively are $T_2$ and $T_1$ ($T_2$ > $T_1$). The rate of heat transfer through the slab, in a steady state is $\left( {\frac{{A({T_2} - {T_1})K}}{x}} \right)f$, with $f $ which equal toView Solution
- 8View SolutionNewton's law of cooling is used in laboratory for the determination of the
- 9An ice box used for keeping eatable cold has a total wall area of $1\;metr{e^2}$ and a wall thickness of $5.0cm$. The thermal conductivity of the ice box is $K = 0.01\;joule/metre{ - ^o}C$. It is filled with ice at ${0^o}C$ along with eatables on a day when the temperature is $30°C$ . The latent heat of fusion of ice is $334 \times {10^3}joules/kg$. The amount of ice melted in one day is ........ $gms$ ($1day = 86,400\;\sec onds$)View Solution
- 10A body takes $5$ minutes for cooling from ${50^o}C$ to ${40^o}C.$ Its temperature comes down to ${33.33^o}C$ in next $5$ minutes. Temperature of surroundings is ....... $^oC$View Solution
