That gas cannot be liquified
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The molar heat capacity in a process of a diatomic gas if it does a work of $\frac{Q}{4}$ when a heat of $Q$ is supplied to it isView Solution
- 2View SolutionTwo moles a monoatomic gas is mixed with six moles of a diatomic gas. The molar specific heat of the mixture at constant volume is:
- 3The pressure and volume of saturated water vapour are $P$ and $V$ respectively. It is compressed isothermally thereby volume becomes $V/2,$ the final pressure will beView Solution
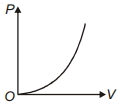
- 4The variation of pressure $P$ with volume $V$ for an ideal diatomic gas is parabolic as shown in the figure. The molar specific heat of the gas during this process isView Solution
- 5The temperature of an ideal gas is reduced from $927^\circ C$ to $27^\circ C$. The $r.m.s.$ velocity of the molecules becomesView Solution
- 6Under an adiabatic process, the volume of an ideal gas gets doubled. Consequently the mean collision time between the gas molecule changes from $\tau_{1}$ to $\tau_{2} .$ If $\frac{C_{p}}{C_{v}}=\gamma$ for this gas then a good estimate for $\frac{\tau_{2}}{\tau_{1}}$ is given by :View Solution
- 7View SolutionMolecules of a gas behave like
- 8An ideal gas of Molar mass $M$ is contained in a vertical tube of height $H$, closed at both ends. The tube is accelerating vertically upwards with acceleration $g$ Then, the ratio of pressure at the bottom and the mid point of the tube will beView Solution
- 9If three moles of monoatomic gas $\left(\gamma=\frac{5}{3}\right)$ is mixed with two moles of a diatomic gas $\left(\gamma=\frac{7}{5}\right)$, the value of adiabatic exponent $\gamma$ for the mixture is:View Solution
- 10A cylindrical container of volume $4.0 \times 10^{-3} \,{m}^{3}$ contains one mole of hydrogen and two moles of carbon dioxide. Assume the temperature of the mixture is $400 \,{K}$ The pressure of the mixture of gases is:View Solution
[Take gas constant as $8.3\, {J} {mol}^{-1} {K}^{-1}$]
