Two moles a monoatomic gas is mixed with six moles of a diatomic gas. The molar specific heat of the mixture at constant volume is:
JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
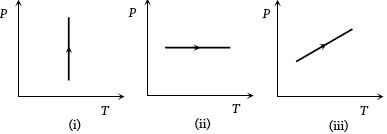
- 2At a given temperature, the pressure of an ideal gas of density $\rho $ is proportional toView Solution
- 3If $\mathrm{n}$ is the number density and $\mathrm{d}$ is the diameter of the molecule, then the average distance covered by a molecule between two successive collisions (i.e. mean free path) is represented by :View Solution
- 4For a given gas at $1\,atm$ pressure, $rms$ speed of the molecules is $200\,m/s$ at $127\,^oC.$ At $2\,atm$ pressure and at $227\,^oC,$ the $rms$ speed of the molecules will beView Solution
- 5One litre of oxygen at a pressure of $1\, atm$ and two litres of nitrogen at a pressure of $0.5\, atm$ are introduced into a vessel of volume $1L$. If there is no change in temperature, the final pressure of the mixture of gas (in $atm$) isView Solution
- 6View SolutionThe temperature below which a gas should be cooled, before it can be liquified by pressure only is termed as
- 7The temperature of an ideal gas at atmospheric pressure is $300\,K$ and volume $1\,m^3$. If temperature and volume become double, then pressure will beView Solution
- 8View SolutionMaxwell distribution function is shown in figure for different gases, which of the following is correct matching?
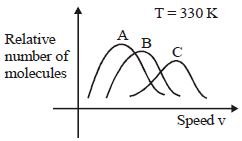
- 9An air bubble doubles its radius on raising from the bottom of water reservoir to be the surface of water in it. If the atmospheric pressure is equal to $10\, m$ of water, the height of water in the reservoir is ..... $m$View Solution
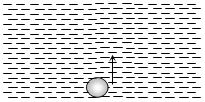
- 10An ideal gas has an initial pressure of $3$ pressure units and an initial volume of $4$ volume units. The table gives the final pressure and volume of the gas (in those same units) in four, processes. Which processes start and end on the same isothermView Solution
$\begin{array}{|c|c|c|c|c|} \hline & A & B & C & D \\ \hline P & 5 & 4 & 12 & 6 \\ \hline V & 7 & 6 & 1 & 3 \\ \hline \end{array}$
