The air density at Mount Everest is less than that at the sea level. It is found by mountaineers that for one trip lasting a few hours, the extra oxygen needed by them corresponds to $30,000\ cc$ at sea level (pressure $1$ atmosphere, temperature $27^oC).$ Assuming that the temperature around Mount Everest is $-73^oC$ and that the oxygen cylinder has capacity of $5.2\ litre,$ the pressure at which $O_2$ be filled (at site) in cylinder is ..... $atm$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$N( < 100)$ molecules of a gas have velocities $1, 2, 3\,.\,.\,.\,.\,.N/km/s$ respectively. ThenView Solution
- 2During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its absolute temperature. The ratio of $\frac{C_p}{C_v}$ for the gas is:View Solution
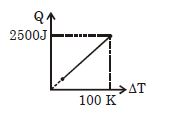
- 3One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
- 4$1$ mole of gas occupies a volume of $100\, ml$ at $50\, mm$ pressure. What is the volume occupied by two moles of gas at $100\, mm$ pressure and at same temperature ..... $ml$View Solution
- 5A vessel contains a mixture of one mole of oxygen and two moles of nitrogen at $300\ K.$ The ratio of the average rotational kinetic energy per $O_2$ molecule to that per $N_2$ molecule isView Solution
- 6Match Column $- I$ and Column $- II$ and choose the correct match from the given choices.View Solution
Column $- I$ Column $- II$ $(A)$ Root mean square speed of gas molecules $(P)$ $\frac{1}{3} \mathrm{n} m \bar{v}^{2}$ $(B)$ Pressure exerted by ideal gas $(Q)$ $\sqrt{\frac{3 \mathrm{RT}}{\mathrm{M}}}$ $(C)$ Average kinetic energy of a molecule $(R)$ $\frac{5}{2} \mathrm{RT}$ $(D)$ Total internal energy of $1$ mole of a diatomic gas $(S)$ $\frac{3}{2} \mathrm{k}_{\mathrm{B}} \mathrm{T}$ - 7A polyatomic ideal gas has $24$ vibrational modes. What is the value of $\gamma$ ?View Solution
- 8An insulated box containing a diatomic gas of molar mass $M$ is moving with a velocity $v$. The box is suddenly stopped. The resulting change in temperature isView Solution
- 9A vessel contains $1$ mole of $O_2$ gas (molar mass $32$) at a temperature $T$. The pressure of the gas is $P$. An identical vessel containing one mole of $He$ gas (molar mass $4$) at a temperature $2T$ has a pressure ofView Solution
- 10$S.I.$ unit of universal gas constant isView Solution
