A vessel contains $1$ mole of $O_2$ gas (molar mass $32$) at a temperature $T$. The pressure of the gas is $P$. An identical vessel containing one mole of $He$ gas (molar mass $4$) at a temperature $2T$ has a pressure of
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionSaturated vapour is compressed to half its volume without any change in temperature, then the pressure will be
- 2Two vessels $A$ and $B$ are of the same size and are at same temperature. A contains $1 \mathrm{~g}$ of hydrogen and $B$ contains $1 \mathrm{~g}$ of oxygen. $\mathrm{P}_{\mathrm{A}}$ and $\mathrm{P}_{\mathrm{B}}$ are the pressures of the gases in $A$ and $\mathrm{B}$ respectively, then $\frac{\mathrm{P}_{\mathrm{A}}}{\mathrm{P}_{\mathrm{B}}}$ is:View Solution
- 3The volume $V$ of an enclosure contains a mixture of three gases, $16\, g$ of oxygen, $28\, g$ of nitrogen and $44\, g$ of carbon dioxide at absolute temperature $T$. Consider $R$ as universal gas constant. The pressure of the mixture of gases isView Solution
- 4Hydrogen gas and oxygen gas have volume $1 \,\,cm^3$ each at $N.T.P$.View Solution
- 5A gas is enclosed in a vessel at a constant temperature at a pressure of $5$ atmosphere and volume $4$ litre. Due to a leakage in the vessel, after some time, the pressure is reduced to $4$ atmosphere. As a result, theView Solution
- 6A spherical soap bubble inside an air chamber at pressure $P_0=10^5 Pa$ has a certain radius so that the excess pressure inside the bubble is $\Delta P=144 Pa$. Now, the chamber pressure is reduced to $8 P_0 / 27$ so that the bubble radius and its excess pressure change. In this process, all the temperatures remain unchanged. Assume air to be an ideal gas and the excess pressure $\Delta P$ in both the cases to be much smaller than the chamber pressure. The new excess pressure $\Delta P$ in $Pa$ isView Solution
- 7The density of a gas at normal pressure and $27°C$ temperature is $24.$ Keeping the pressure constant, the density at $127°C$ will beView Solution
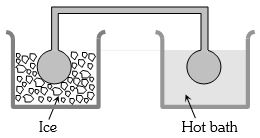
- 8Two identical glass bulbs are interconnected by a thin glass tube. A gas is filled in these bulbs at $N.T.P.$ If one bulb is placed in ice and another bulb is placed in hot bath, then the pressure of the gas becomes $1.5$ times. The temperature of hot bath will be ....... $^oC$View Solution
- 9Consider a gas with density $\rho $ and $\bar c$ as the root mean square velocity of its molecules contained in a volume. If the system moves as whole with velocity $v,$ then the pressure exerted by the gas isView Solution
- 10An open and wide glass tube is immersed vertically in mercury in such a way that length $0.05\,\, m$ extends above mercury level. The open end of the tube is closed and the tube is raised further by $0.43 \,\,m$. The length of air column above mercury level in the tube will be ...... $m$ Take $P_{atm} = 76 \,\,cm$ of mercuryView Solution
