Match Column $- I$ and Column $- II$ and choose the correct match from the given choices.
| Column $- I$ | Column $- II$ |
| $(A)$ Root mean square speed of gas molecules | $(P)$ $\frac{1}{3} \mathrm{n} m \bar{v}^{2}$ |
| $(B)$ Pressure exerted by ideal gas | $(Q)$ $\sqrt{\frac{3 \mathrm{RT}}{\mathrm{M}}}$ |
| $(C)$ Average kinetic energy of a molecule | $(R)$ $\frac{5}{2} \mathrm{RT}$ |
| $(D)$ Total internal energy of $1$ mole of a diatomic gas | $(S)$ $\frac{3}{2} \mathrm{k}_{\mathrm{B}} \mathrm{T}$ |
NEET 2021, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The molar specific heats of an ideal gas at constant pressure and volume are denoted by $C _{ P }$ and $C _{ V }$ respectively. If $\gamma=\frac{C_{p}}{C_{V}}$ and $R$ is the universal gas constant, then $C _{ V }$ is equal toView Solution
- 2When pressure remaining constant, at what temperature will the $r.m.s.$ speed of a gas molecules increase by $10 \%$ of the r.m.s. speed at $STP$?View Solution
- 3When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will beView Solution
- 4Graph between volume and temperature for a gas is shown in figure. If $\alpha$ = Volume coefficient of gas = $\frac{1}{273}$ per $^o C$,then what is the volume of gas at a temperature of $819 ^o C$View Solution
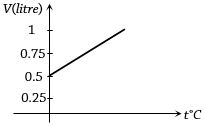
- 5$1$ mole of gas occupies a volume of $100\, ml$ at $50\, mm$ pressure. What is the volume occupied by two moles of gas at $100\, mm$ pressure and at same temperature ..... $ml$View Solution
- 6$N\,moles$ of a diatomic gas in a cylinder are at a temperature $T$. Heat is supplied to the cylinder such that the temperature remains constant but $n\,moles$ of the diatomic gas get converted into monoatomic gas. What is the change in the total kinetic energy of the gas ?View Solution
- 7View SolutionThe molar specific heat at constant pressure for a monoatomic gas is
- 8Pressure $Vs$ temperature graph of an ideal gas is shown in figure. Density of the gas at point $A$ is $\rho _0$ , Density at $B$ will beView Solution
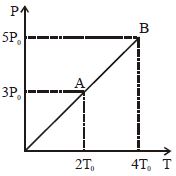
- 9The figure shows the volume $V$ versus temperature $T$ graphs for a certain mass of a perfect gas at two constant pressures of ${P_1}$ and ${P_2}$. What interference can you draw from the graphsView Solution
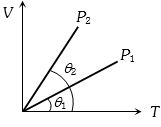
- 10Statement$-1$ : Real gas approaches ideal gas behaviour for low pressures and high temperatures.View Solution
statement$-2 $: At low pressure, density of gas is very low.
