$S.I.$ unit of universal gas constant is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA diatomic molecule has how many degrees of freedom
- 2In case of hydrogen and oxygen at $N.T.P.$, which of the following quantities is $/$ are the same?View Solution
- 3View SolutionAt constant volume, for different diatomic gases the molar specific heat is
- 4If a given mass of gas occupies a volume of $10\, cc$ at $1$ atmospheric pressure and temperature of $100°C(373.15\, K).$ What will be its volume at 4 atmospheric pressure; the temperature being the same .... $cc$View Solution
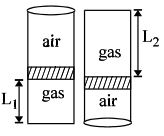
- 5An ideal gas is trapped inside a test tube of cross-sectional area $20 \times 10^{-6} \,\,m^2$ as shown in the figure. The gas occupies a height $L_1$ at the bottom of the tube and is separated from air at atmospheric pressure by a mercury column of mass $0.002\,\, kg$. If the tube is quickly turned isothermally, upside down so that $L_2$ mercury column encloses the gas from below. The gas now occupies height $L_1$ in the tube. The ratio $L_1$ is [Take atmospheric pressure $= 10^5 Nm^{-2}]$View Solution
- 6The gas in vessel is subjected to a pressure of $20$ atmosphere at a temperature $27°C.$ The pressure of the gas in a vessel after one half of the gas is released from the vessel and the temperature of the remainder is raised by $50°C$ is ....... $atm$View Solution
- 7View SolutionThe number of translational degrees of freedom for a diatomic gas is
- 8The density of a gas at normal pressure and $27°C$ temperature is $24.$ Keeping the pressure constant, the density at $127°C$ will beView Solution
- 9A cubical box with porous walls containing an equal number of ${O_2}$ and $H_2$ molecules is placed in a large evacuated chamber. The entire system is maintained at constant temperature $T.$ The ratio of ${v_{rms}}$ of ${O_2}$ molecules to that of the ${v_{rms}}$ of $H_2$ molecules, found in the chamber outside the box after a short interval isView Solution
- 10For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.View Solution
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$