The average kinetic energy of a helium atom at ${30^o}C$ is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In an ideal gas at temperature $T,$ the average force that a molecule applies on the walls of a closed container depends on $T$ as $T^q$ . A good estimate for $q$ isView Solution
- 2One mole of an ideal gas at $STP$ is heated in an insulated closed container until the average speed of its molecules is doubled. Its pressure would therefore increase by factor.View Solution
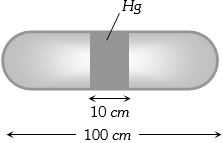
- 3A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 4In the relation $n = \frac{{PV}}{{RT}},n = $View Solution
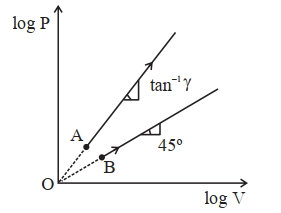
- 5Two thermodynamical process are shown in the figure. The molar heat capacity for process $A$ and $B$ are $C_A$ and $C_B$. The molar heat capacity at constant pressure and constant volume are represented by $C_P$ and $C_V$, respectively. Choose the correct statement.View Solution
- 6If three moles of monoatomic gas $\left(\gamma=\frac{5}{3}\right)$ is mixed with two moles of a diatomic gas $\left(\gamma=\frac{7}{5}\right)$, the value of adiabatic exponent $\gamma$ for the mixture is:View Solution
- 7Consider an ideal gas confined in an isolated closed chamber. As the gas undergoes an adiabatic expansion, the average time of collision between molecules increases as $V ^q$, where $V$ is the volume of the gas. The value of $q$ is $\left( {\gamma = \frac{{{C_P}}}{{{C_V}}}} \right)$View Solution
- 8When the temperature of a gas is raised from $27^o C$ to $90^o C$, the percentage increase in the $r.m.s.$ velocity of the molecules will be ..... $\%$View Solution
- 9The density of a gas at normal pressure and $27°C$ temperature is $24.$ Keeping the pressure constant, the density at $127°C$ will beView Solution
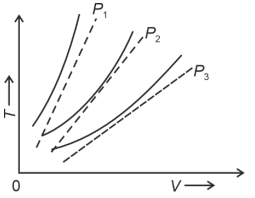
- 10The following graph represents the $T-V$ curves of an ideal gas (where $T$ is the temperature and $V$ the volume) at three pressures $P_1, P_2$ and $P_3$ compared with those of Charles's law represented as dotted lines.View Solution
Then the correct relation is: