The efficiency of a Carnot engine operating with reservoir temperature of $100\,^{\circ} C$ and $-23\,^{\circ} C$ will be
AIPMT 1997, Easy
Reservoir temperature $\left(T_{1}\right)=100^{\circ} C =373$ $K$ and $T_{2}=-23^{\circ} C =250 K$
The efficiency of a Carnot engine
$\eta=\frac{T_{1}-T_{2}}{T_{1}}=\frac{373-250}{373}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
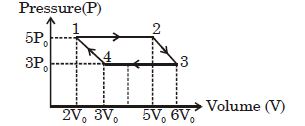
- 1An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
- 2A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 3A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
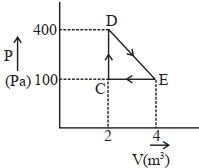
- 4View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 5Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 6The temperature of reservoir of Carnot's engine operating with an efficiency of $70\%$ is $1000K.$ The temperature of its sink is ...... $K$View Solution
- 7$0.02\, moles$ of an ideal diatomic gas with initial temperature $20^{\circ} C$ is compressed from $1500 \,cm ^{3}$ to $500 \,cm ^{3}$. The thermodynamic process is such that $p V^{2}=\beta$, where $\beta$ is a constant. Then, the value of $\beta$ is close to (the gas constant, $R=8.31 \,J / K / mol$ ).View Solution
- 8Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
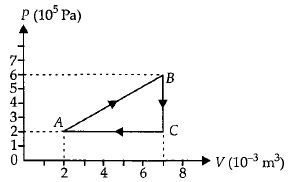
- 9A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 10$2$ moles of a monoatomic gas are expanded to double its initial volume, through a process $P/V =$ constant. If its initial temperature is $300\,\, K$, then which of the following is not true.View Solution
